Locus Biosciences is a precision medicine company that is developing bacteriophage-based products to precisely remove pathogenic bacteria from the human body and to leverage the microbiome to treat diseases across multiple therapeutic areas.
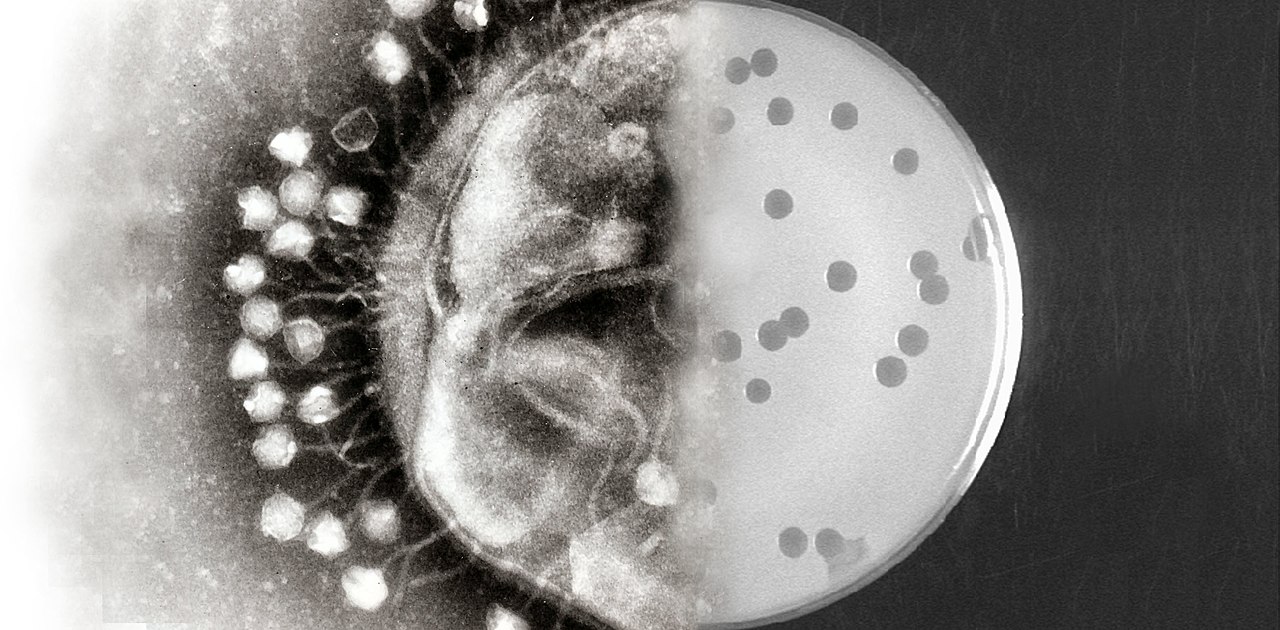
Bacteriophage therapy, also known as phage therapy, is a promising alternative to antibiotics for the treatment of bacterial infections. Bacteriophages are viruses that specifically target and infect bacteria, replicating inside the bacterial cell until it bursts and releases new phages.
Phage therapy has several advantages over antibiotics, including greater specificity, fewer side effects, and the ability to evolve alongside bacteria, reducing the risk of resistance.
While phage therapy has been used in some countries for several decades, it is now gaining renewed interest and investment as antibiotic resistance becomes an increasingly pressing global health threat.
Locus Biosciences CEO Paul Garofolo discussed the ways in which his company is maximizing bacteriophages potential as antibacterial drugs and also as gene-delivery vectors. (The interview has been edited for length and clarity.)
What was it about phages that attracted you to them to work with as a therapeutic?
I was at North Carolina State University working with an entrepreneurship class – the TEC program – as an executive in residence. A group of students brought the CRISPR-Cas3 technology with the goal of finding a way to commercialize the technology but they didn’t really have a delivery vector. Together, we started looking at lots of different delivery vectors like cell penetrating peptides and nanoparticles. Bacteriophage (phage for short) was one of the possible carriers for the technology at the time.
Phages are attractive as delivery vectors because of their safety profiles, and the size of their genomes. When you look at those two things together, they make an incredibly ideal viral vector for delivering a therapeutic payload into the human body.
Human viruses attack human cells; bacteriophage only attack bacterial cells. With phage, you don’t have to worry that whatever payload you’re working with is going to be injected into human cells. That creates a safety profile that is super unique.
The average genome sizes of phages that we work with typically range from 60,000 to more than 120,000 base pairs. We found that the general rule is that you can usually get a deletion or an insertion to hold while manipulating less than 10% of the genome size of the virus. It gives you a sizable chunk – something between 8,000 and maybe even up to 12,000 base pairs of size – that you can manipulate for delivering a therapeutic payload.
While our roots started with CRISPR, we’ve really expanded out from there and now we are looking at all different kinds of payloads that are optimized for different purposes.
Why are engineered phages attractive as an antibacterial therapy?
Phages have evolved over billions of years with bacteria to form an ecological balance, both in nature and in the human body. Estimates vary anywhere from 40% to more than 70% of the cells that are in or on the human body at any time are bacteria, mostly in the gastrointestinal tract. There are phages in the water we drink, in the salads and other food we eat, pretty much everywhere – that we are exposed to phages all of the time because phages exist everywhere that bacteria are found.
Phages have historically been compared to antibiotics, which decades ago were well into the high 90%s in terms of efficacy and were very cheap to manufacture and deliver. Antibiotic resistance was much less of a problem back then. Nowadays, with multidrug resistant bacteria running rampant, efficacy rates for antibiotics are dramatically dropping. We have also come to better appreciate the negative side effects of blasting your microbiome with broad-spectrum antibiotics and indiscriminately killing a large portion of the healthy bacteria in your body. It’s an issue becoming a lot more relevant in today’s medicine.
I think we’ve been learning over decades of use of antibiotics that they’re not going to last forever and they can be highly toxic and damaging to the human body, especially when administered over long periods of time. We don’t really know what that long term impact is to the human body except for the possible lack of efficacy over time, antibiotic resistance growth, as well as potentially side effects to comorbid patients that are life threatening.
This creates this window of opportunity to say, “Okay, well, maybe we revisit phages. How can you work with that? How can you work with a more specific biological approach rather than a small molecule approach to take bacteria out of the body that you don’t want there while leaving the healthy bacteria unharmed?”
When we started Locus Biosciences, it was super exciting. Can we build a therapy platform – not a single therapy but rather a platform – that could systematically create precision bacterial removal drugs that just would go into the human body and take out just the species you were worried about? Could we do it at levels that were comparable to antibiotics?
What we’ve been finding is that yes, we can engineer these phages to carry payloads that help increase their natural killing efficacy. If we can do that, we very well could produce a new drug modality that one day could complement or even replace older antibiotics. That’s certainly the hunt that we’re on and we’ve got very encouraging results in animal studies.
We just started our statistically powered, proof-of-concept Phase 2/3 trial. We hope that within 18 months, we’ll have gotten the evidence of efficacy in humans that the modality works as we expect and that we’re able to eradicate targeted bacteria very specifically.
So, what technologies in particular have changed the way Locus is working with phages? What would you say has been very important in terms of technological advancements?
During the last 10 years, the commoditization of DNA sequencing – being able to sequence the genome of a bacterium or a phage for a reasonable amount of money – and the ability to deeply understand the bacteria that you’re trying to kill as well as the phages that could do it. That is definitely a big one. There’s been an opening up of understanding of what a lot of those genes are actually doing in bacteria and phages. Twenty or 30 years ago, it just wasn’t financially something that could be accomplished and now we do it routinely every day.
The other great technological advance has been robotics, being able to use high throughput liquid handlers to be able to, en masse, find the phages and be able to think through how to put them together in cocktails. This makes it possible to create “commercializable” drugs that do not need to be customized for the needs of each individual patient.
Then without question, bioinformatics. The whole rise of computational genomics, being able to handle all of the data that’s coming off of these high throughput runs, to be able to crunch it and understand how we create drugs that are comparable to antibiotics.
Those three things opened up the door to be able to do synthetic biology at scale for a reasonable cost.
Ultimately, engineering viruses is the new frontier of medicine. It’s not just in our space, but also gene therapy and related fields. Being able to use a virus and engineer its payload to edit genes or remove a target — that is the new horizon in medicine.
What is CRISPR phage and how are trials going?
CRISPR phage is basically just taking CRISPR enzymes and engineering them as a payload within the bacteriophage to enhance its bactericidal effect relative to the natural phage. We make fixed cocktails of bacteriophage that kill the vast majority of the clinically-relevant strains of the target bacteria. We create a CRISPR cassette which we engineer into the genomes of the phage that are in that cocktail.
We’re working with a lot more than CRISPR. These days, we’re working with enzymes that degrade biofilms, various types of bactericidal molecules, and other payloads in addition to CRISPR systems such as CRISPR-Cas3. Then we load our payloads into any of the genomes in that cocktail that are of enough size that they can carry that payload. That may then become our fixed drug product. Then that product is moved into clinical trials.
Bacteriophages are part of the natural ecosystem and what we found — and what I think is the fundamental limitation of phage alone as a wild type — is that it will not kill all of its food. It’s a virus. If it eradicates its food, it’s dead. Instead you’ll have a bacteria that begins to sort of grow at an uncontrolled pace like an infection and the phages come in and it brings it back down to some equilibrium, some balance. Our approach to engineering the phage disrupts that balance and allows the phage to clear the infection.
In terms of clinical trials, we’ve completed our first ever well-controlled, double-blind clinical trial. It’s the first of its kind where we had double blinded study for safety and tolerability. We completed that trial in 2021 and it gave us the safety and tolerability profile data that we needed to be able to move into our current Phase 2/3 clinical trial. We’re specifically trying to go after a patient population that has urinary tract infections that are caused by multi-drug resistant strains of E. coli.
Why are cocktails of phages the most effective way to move forward?
You can’t find one phage that kills all the strains of a species, let’s say Klebsiela pneumoniae. What we do instead is put several phages together into a cocktail to try to get a fixed set of phages to kill above 95% of all known strains of that species. The way we do that is we look temporally and geographically to create a panel of bacterial isolates from a contemporary set of patients. We look for things that are recent, within the last two to four years, for example.
We screen multiple times until we know we have the smallest block of phage that hits our 95% target that we believe gives you the ability to have susceptibility comparable to an antibiotic.
Where do you get your phages?
We have found that the highest density of novel phages come from wastewater treatment plants that are in strategic positions. If you can get to a wastewater treatment plant that’s coming off an international airport, you’re probably picking up a lot of really good phages.
It’s interesting. We do find a lot of the same phages regardless of what geographic region of the world you’re in. It’s not that every bacteria works the same but we’ve mined through tons of samples. We find that phages from Asia and Europe are similar to what’s coming out of the United States. We’re becoming a smaller place with international travel.
We’ve been able to find phage for the vast majority of bacterial targets we’ve ever tried to go after, including some that had no phages reported in the literature at all. We’re pretty good at finding them, including from soil samples from farms across the country, livestock samples and human body sampling. I think wastewater probably represents the highest producers.
Can you discuss the using page to potentially program bacteria the potential for him to have them as sort of little biologics makers inside the body or wherever?
That’s a cool idea we’ve been exploring. There’s a lot of bacteria that have persistently colonized the body. There are also bacteria that are transient. If you if you put a payload – let’s say nanobody – into a phage capsid and you send it in to go after a colonized bacteria – so something that’s always in there – you haven’t engineered it to eradicate those bacteria. Instead, you’ve just engineered it to keep that natural stasis and amplify some amount, producing a payload that has a biological effect that you’re interested in, right where you want it.
You’ve built your facility from scratch. Can we discuss why that’s important?
I used to be the chief technology officer at Pantheon and ran worldwide operations for all clinical trial material manufacturing under contract for roughly 450 different experimental compounds. I knew a lot about manufacturing.
Phages are super contagious among their bacterial hosts. When you’re running a manufacturing facility, phage are something that you try to avoid. If you ever “phaged out”, it’ll shut down your facility for a long time while you remediate it and then prove that it’s gone. I knew that coming into this business. That was one of the first things I thought of. I was like, nobody’s going to let us manufacture this thing in a facility that uses bacterial cells to manufacture other products. We really didn’t have a choice but to build the manufacturing site.
When we first started the company, we just converted a small little lab at an innovation center and put the controls in place to produce our phase one material. Off of that success, we knew we needed a viral vector production core, so we built an awesome one. Building out a manufacturing facility as an early stage company is not for the faint hearted, but we did a great job at it.
We won the ISP Facility of the Year Award last year, Honorable Mention category and I think that’s not just because it’s not a big site — It’s 10,000 square feet – but also because it’s ready for commercial launch.
IMAGE CREDIT: Emily Brown.




Leave a Reply