A gene (PIEZO1) for ion channels that govern cellular responses to mechanical stimuli may be a potential target for therapies for musculoskeletal disorders. Cells that are activated by mechanical stress (i.e., mechanosensitive) allow, for example, the sensation of touch at the fingertips. In bones, muscles, and tendons, mechanosensitive cells also play an important role—by initiating the production or breakdown of tissue in response to physical demands. New findings show that a significant biomechanical advantage is ppimparted by increased mechanosensitivity in tendons.
- Early complex life clung to oxygenated seafloors for hundreds of millions of years
- The fungus that spoils nearly everything
- New tool identifies blackouts during drinking
- Beyond Mendel: New Study Reveals the Startling Complexity of Epigenetic Inheritance
- AI atlas reveals hidden whole-body-damage caused by obesity
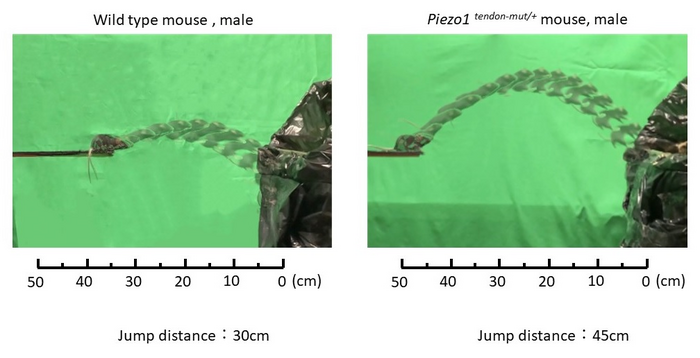
In a comprehensive study published this month in Science Translational Medicine, researchers at TMDU, Scripps Research, and others found that a genetic variant (R2482H Piezo1) in mice, which is analogous to one (E756del PIEZO1) in humans, produced cellular changes in tendons that translated to altered tissue properties and physical characteristics, which in turn translated to better athletic ability.
Both the human genetic variant and the mouse genetic variant were known to impart resistance to malaria (by altering the shape of red blood cells) and were associated with better tendon properties in mice and better jumping ability in humans. However, the link between genetic expression and physical performance was unclear.
HAVE YOUR SAY.
Join us in The Bullpen, where the members of the Scientific Inquirer community get to shape the site’s editorial decision making. We’ll be discussing people and companies to profile on the site. On Wednesday June 22nd at 5:30pm EST, join us on Discord and let’s build the best Scientific Inquirer possible.
The research team compared four groups of mice: wild-type mice (with no modifications to gene expression) and mice with tendon-specific, muscle-specific, or whole-body expression of the genetic variant. “Tendons were able to stretch more easily and store larger amounts of energy, which allowed for greater flexion and greater instantaneous power produced by the ankle, whether alterations were innate or introduced after mice matured,” explains senior author Hiroshi Asahara. “Muscle-specific alterations, however, did not result in improved physical performance.”
High expression of the calcium-ion channel mechanoreceptor coincided with high expression of two transcription factors, found only in tendon cells, that regulate genes for collagen and proteoglycan production.
Lead author Ryo Nakamichi explains, “Given these findings, we looked at tendon structure and composition more closely. Increased mechanosensitivity, from either tendon-specific or whole-body alterations, resulted in wider tendons composed of collagen fibrils with larger diameters.”
To put the findings from mice into human context, the research team replicated the effect of PIEZO1 activation on expression of the same two transcription factors in human tendon and, in addition, compared the frequency of the E756del PIEZO1 genetic variant in elite sprinters with that of nonathletes in Jamaica (because the variant occurs in a larger proportion in populations of African descent). The frequency of E756del PIEZO1 was significantly higher in the sprinters.
This finding that the specific variant of PIEZO1 gene can potentiate physical activity may eventually allow for new treatments for individuals with tendinopathy or other tendon-related disorders to be developed.
IMAGE CREDIT: Department of Systems BioMedicine, TMDU


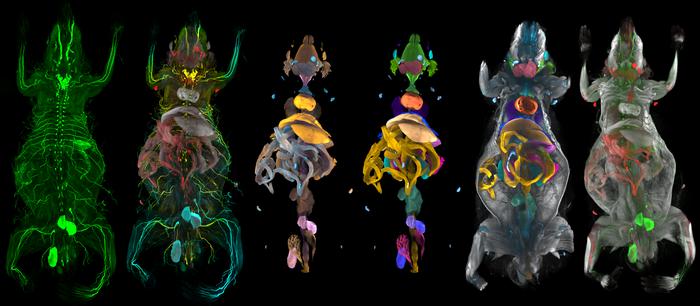
Leave a Reply