Chloroplasts are essential to plant life. They are found in every plant cell. Photosynthesis occurs within the organelles’ interior, converting sunlight into usable energy stored in the form of ATP and NADP. That energy gradually makes its way through the entire food chain. To say that understanding and possibly manipulating chloroplasts is important is an understatement.
Central to the way chloroplasts work are the DNA (cpDNA) contained inside, genetic material distinct from nuclear DNA. Once belonging to a separate organism that was incorporated long ago, it holds the key to gaining better control of how a plant functions.
Researcher Nishimura Yoshiki from Kyoto University in Japan has been studying cpDNA for over twenty years. He recently published a paper describing his findings regarding a gene called moc1 that has befuddled researchers for decades. It also goes on to describe the unexpected discovery of Holliday Junctions within cpDNA. He set aside time from his research schedule to chat with SCINQ.
SCIENTIFIC INQUIRER: What prompted you to study nucleoid segregation in chloroplasts?
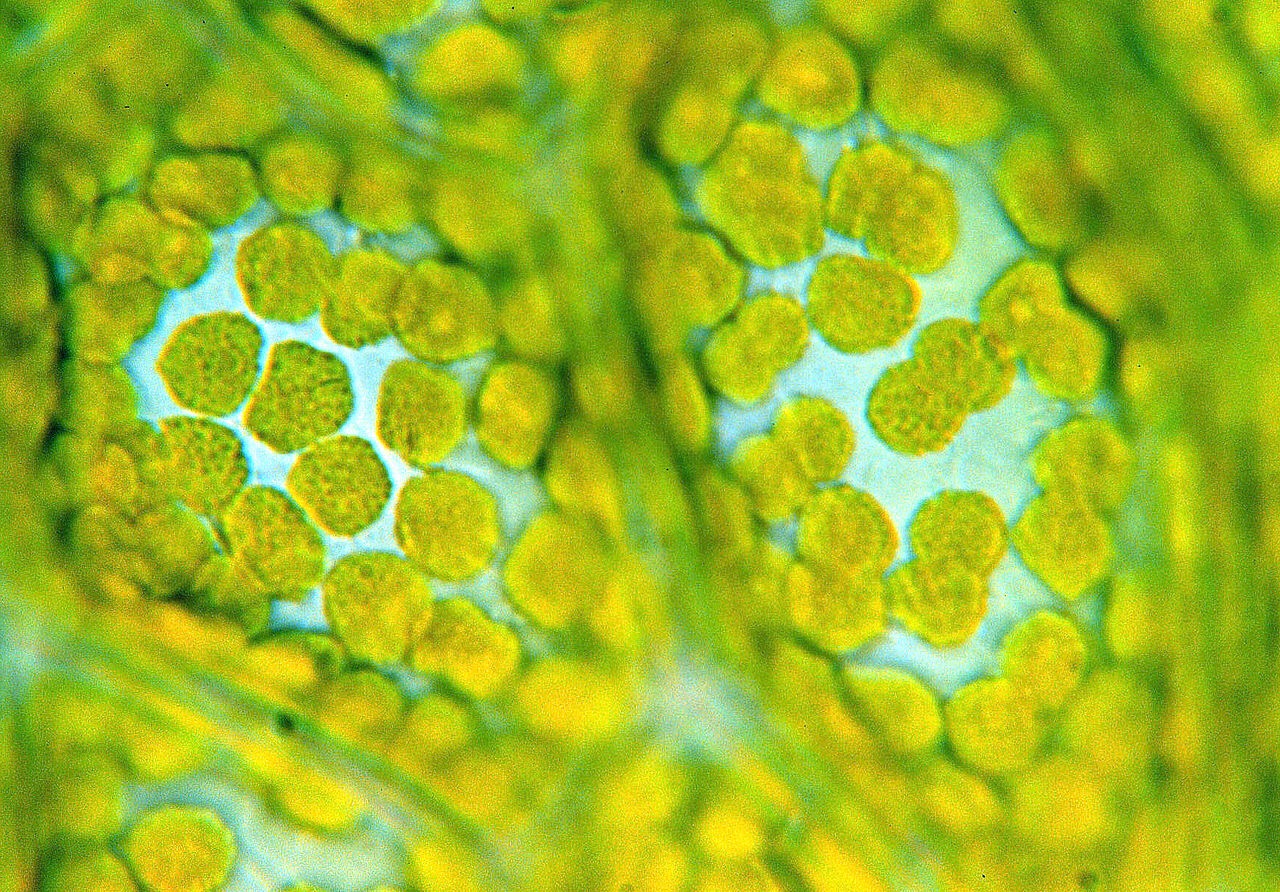
NISHIMURA YOSHIKI: The faithful maintenance and inheritance of DNA molecules is a fundamental issue for all living organisms. Chloroplasts possess multiple copies (~80-100 copies) of their own DNA called chloroplast DNA – cpDNA – that encode essential components for photosynthesis. cpDNA molecules are packaged into cpDNA-protein complexes known as chloroplast nucleoids. Chloroplast nucleoids are thought to be a functional platform for cpDNA replication, repair, gene expression, inheritance, etc. similar to the chromosomes in the cell nucleus.
Chloroplast nucleoids are minute dot-like structures (< 1 μm) within chloroplasts, but their behaviors are surprisingly dynamic. Their shape, number and distribution change markedly depending on the cell cycle, developmental stage, nutritional environment, and species of green plants. Obviously there is an evolutionary difference. However, the detailed molecular systems that underlie the dynamism of chloroplast nucleoids have remained elusive. So we decided to solve this problem.
SCINQ: How did you design the experiment and what was your objective?
NY: For the purpose described above, we designed our screening strategy to isolatemutants of the organism chlamydomonas, where they had defective morphology of chloroplast nucleoids. Our strategy was simple; we observe each strain one by one under the microscope. After observing thousands of strains, several mutants were isolated. The mutants had an irregularly aggregated cp nucleoid and so it was designated as monokaryotic chloroplasts (moc). The moc mutants were reported in 1999 but the analysis of the mutants were troublesome and did not go well.
SCINQ: Why did you focus on moc1 and what did you discover about the gene?
NY: The analysis of moc1 was so troublesome back in 1999, partly because of the lack of chlamydomonas genome information.
With the latest information of chlamydomonas genome, and new experimental technologies, we finally identified the gene responsible for the moc phenotype after – 20 years. Initially, to our disappointment, BLAST search did not detect any homologues. However, SWISS modeling detected a structural homology between our gene product and bacterial Holliday junction resolvase, RuvC.
SCINQ: What is the significance of discovering Holliday junctions (HJ) in chloroplasts?
NY: HJ resolvase is an endonuclease that cuts the Holliday junction, a four-way DNA structure, in a sequence/structure-specific manner to finalize the process of homologous recombination. This enzyme is so critical for DNA metabolism that it has been identified in bacteria, the eukaryotic cell nucleus (Wu et al., Nature 2003; Boddy et al., Cell 2001; Ip et al., 2008 Nature), and mitochondria (Kleff et al., EMBO J. 1992; Lockshon et al., Cell 1995), but until now HJ resolvase in chloroplasts has remained unidentified.
Homologous recombination plays a crucial role in the replication and repair of DNA molecules and it would be especially important in chloroplasts where DNA damaging ROS (reactive oxygen species) would be produced due to the photosynthetic electron flow. Therefore, without HJ resolvase, HJs of cpDNA molecules cannot be resolved and remain entangled, resulting in unequal inheritance of cpDNAs, and ultimately death of individual plants.
SCINQ: How did you go about visualizing moc1 activity?
NY: When we obtained the biochemical data to demonstrate the HJR activity of moc1, we felt that it would be ideal if we could have other supporting data using different technology and methodology.
We were very lucky to find a wonderful collaborator familiar with DNA origami and Atomic force microscopy in the same building as our laboratory. So we analyze our freshly prepared recombinant proteins with their cutting-edge technology.
SCINQ: What are the broader implications of your discovery and studying chloroplasts, in general?
NY: Homologous recombination is critical for the repair of cpDNA molecules so it may be possible to produce plants with higher resistance against stressful conditions that would damage cpDNA molecules.
Also, cpDNAs are target of genetic engineering to produce beneficial proteins, such as vaccines. The genetic engineering of cpDNA, what we call chloroplast transformation, is based on the efficient homologous recombination. However, currently the technique is available for limited plant species, such as tobacco, rice and some algae. By understanding and improving the mechanisms of homologous recombination in chloroplasts, it may be possible to achieve chloroplast transformation in other plant species.
SCINQ: On a personal level, what brought you to a life in the sciences? In particular plant biology.
NY: The origin and evolution of life. The endosymbiotic theory, in particular, is the most attractive subject for me.
In that context, the evolution from algae to land plants is very interesting. We can find not only mitochondria but also plastids and many dynamic events such as secondary and tertiary endosymbiosis.
SCINQ: Can you place this experiment into your past work? How does it fit in? How does it influence future investigations?
NY: We’ve been working on moc mutant for the last 20 years and now we feel relieved to have completed the project.
We are also very excited because the analysis of moc mutants lead to the serendipitous discovery of HJR in chloroplasts.
This will be a new starting point for our further studies on the molecular mechanism of homologous recombination in chloroplasts.
SCINQ: What role do you believe the scientist play in the world?
NY: To stay curious for the unknown world
SCINQ: Finally, what would you be if you were not a scientist?
NY: I once dreamed of working for the United Nations.
*
For more information about Nishimura Yoshiki visit his lab page. For more about his work on moc1.
For background information about chloroplasts:
For background information about Holliday Junctions:
SCINQ is looking for contributors in all shapes and sizes. Columns, blogs, long form, short form, interviews, what have you. Topics can range from hard science, soft science, sports, movies, books, and music. If it marries science and life, we’re interested. Use your imagination. [Learn more]




Leave a Reply