In October 2024, Teva Pharmaceuticals USA initiated a nationwide recall of Prazosin hydrochloride capsules (1mg, 2mg, and 5mg strengths) due to elevated levels of a carcinogenic impurity called N-nitroso Prazosin C. While the health risk is considered low, the FDA classified this as a Class II recall, and patients are advised to replace their medication and consult with their healthcare provider.
What Is Prazosin HCl?
Prazosin hydrochloride is a medication primarily prescribed to treat high blood pressure (hypertension). It belongs to a class of drugs called alpha-blockers, which work by relaxing and widening blood vessels, making it easier for blood to flow through them and reducing the workload on the heart. This helps lower blood pressure and reduces the risk of stroke, heart attack, and kidney problems.
Beyond hypertension, prazosin is sometimes prescribed off-label for other conditions, including nightmares associated with post-traumatic stress disorder (PTSD) and symptoms of an enlarged prostate.
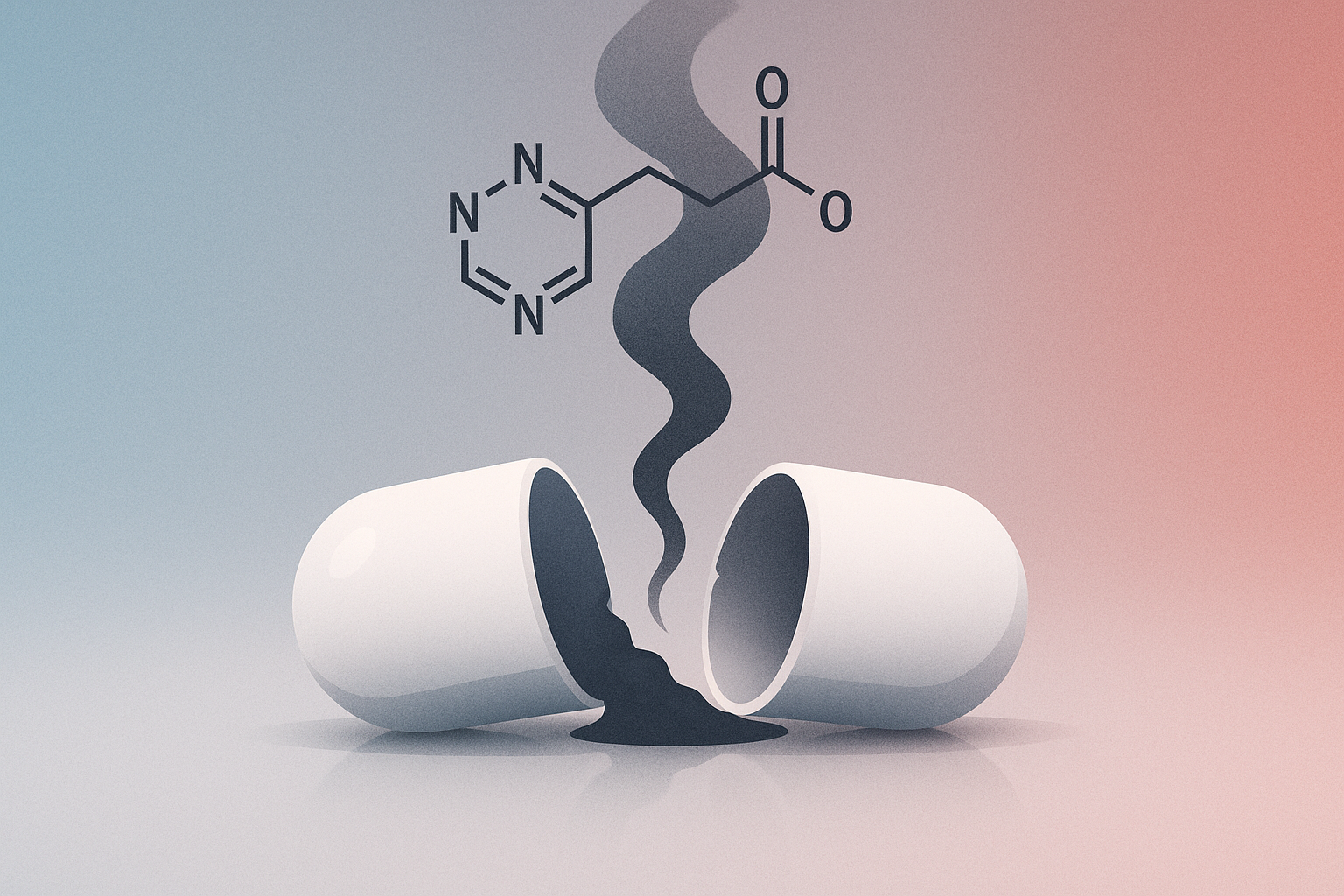
What Is N-Nitroso Prazosin C?
N-nitroso Prazosin C is a nitrosamine impurity—a type of chemical compound that can form during the manufacturing process of certain medications. Nitrosamines are part of a broader family of N-nitroso compounds that have been found in various pharmaceutical products in recent years, prompting increased regulatory scrutiny and testing requirements.
This particular impurity is not an intended ingredient in prazosin but rather an unwanted byproduct that formed during production when the medication did not meet proper manufacturing quality standards.

Why Is It Considered a Carcinogen?
N-nitroso compounds, including N-nitroso Prazosin C, are classified as probable human carcinogens based on extensive scientific research. Here’s why they raise concerns:
DNA Damage: Nitrosamines can interact with DNA in cells, causing mutations that may lead to uncontrolled cell growth—the hallmark of cancer. These compounds can chemically modify DNA structure, potentially triggering cancerous changes over time.
Animal Studies: Laboratory studies in animals have demonstrated that long-term exposure to various nitrosamines can cause tumors in multiple organs, providing evidence for their carcinogenic potential.
Dose and Duration Matter: The cancer risk from nitrosamines depends heavily on the level of exposure and how long someone is exposed. Low levels for short periods pose minimal risk, but higher concentrations over extended periods increase the potential for harm.
The FDA and international regulatory agencies have established acceptable daily intake limits for nitrosamine impurities using the Carcinogenic Potency Categorization Approach (CPCA). This framework sets safety thresholds based on the likelihood that a substance could cause cancer with long-term use. The recalled prazosin capsules exceeded these established safety limits, which is why the recall was necessary.
What Should You Do?
If you’re currently taking prazosin hydrochloride, check your bottle’s lot number and expiration date against the FDA recall list. The recalled bottles (containing 100, 250, 500, or 1,000 capsules) have expiration dates between October 2025 and February 2027.
Do not stop taking your medication abruptly without medical guidance, as this could cause your blood pressure to spike. Instead, contact your healthcare provider immediately to discuss alternative treatment options or replacement medication. Return the recalled medication to your pharmacy for proper disposal and replacement.
For questions about the recall, contact the FDA at 1-888-INFO-FDA (1-888-463-6332).




Leave a Reply