A skull is not one single bone but rather a collection of bone plates joined together early in development. In a study of mice, scientists at the Icahn School of Medicine at Mount Sinai showed how the activity of one gene, turned on in a newly discovered group of bone-bordering cells, may play an important role in shaping the skull. The skulls of mice that were missing the gene were misshapen and were depleted of the cells in a manner that is reminiscent of craniosynostosis, a developmental disorder that affects about one out every 2,500 babies born in the United States.
The lead author of the study was Greg Holmes, PhD, Assistant Professor of Genetics and Genomic Sciences at Icahn Mount Sinai.
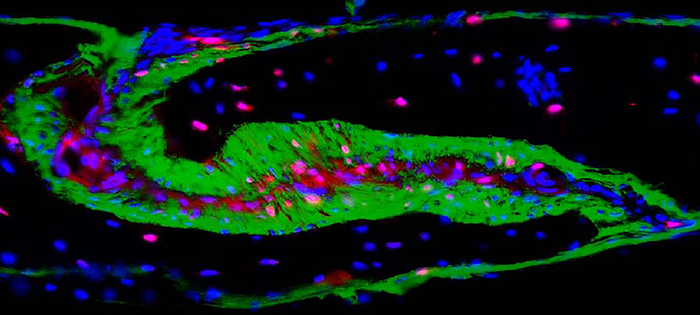
The study focused on the cells of the coronal suture, a fibrous joint that connects the front and middle bone plates. In humans, it runs across the top of the skull, from one temple to the other. Throughout development, the suture is filled with a buffering tissue called mesenchyme, which contains stem cells. The stem cells provide a constant source of new bone cells, or osteoblasts, which are needed as the plates on either side grow and the skull expands. In adults, these stem cells are depleted and the joints between the bone plates fuse.
Craniosynostosis is a birth defect in which this fusion happens prematurely, resulting in babies born with misshapen skulls who may develop neurological problems. Scientists have shown that about 25 percent of cases are linked to genetic mutations and that normal suture development is controlled by a variety of growth factors. Of all the sutures needed to form a skull, the coronal suture is commonly affected in craniosynostosis.
In this study, the Holmes lab worked with researchers in the labs of Bin Zhang, PhD, Harm van Bakel, PhD, and Ethylin Wang Jabs, MD, of Icahn Mount Sinai. Together they studied how the genetic activity in the cells of the coronal suture changes during early development. To do this, they measured the RNA levels of individual suture cells from embryonic mice about one to three days before the mice were normally born.
Their results suggested that a gene encoding a molecule called hedgehog interacting protein (HHIP) plays a unique and critical role in coronal suture development. The gene was more active in a novel group of mesenchyme cells than it was in osteoblasts. In fact, the scientists saw the opposite trend when they looked at the cells of other sutures. Tracing experiments suggested that after birth the coronal suture osteoblasts were derived from these mesenchyme cells. Moreover, the skulls of embryonic mutant mice that were missing the HHIP gene were shaped differently than those from normal mice. Specifically, there were fewer mesenchymal cells separating the skull bones and the mutant coronal suture was close to fusing.
Hedgehog proteins are growth factors known to guide normal growth and development in a variety of species, including the promotion of bone growth. HHIP is known to inhibit hedgehog activity. To the authors of this study, their results suggest that the HHIP gene reduced hedgehog activity to allow normal development of the coronal suture. They hope that advanced single-cell genetic studies like this one will give researchers a more thorough understanding of how a skull is shaped under healthy and disease conditions.
IMAGE CREDIT: Holmes lab, Icahn School of Medicine, NY, NY.





Leave a Reply