Words matter. Images matter. The Scientific Inquirer needs your support. Help us pay our contributors for their hard work. Visit our Patreon page and discover ways that you can make a difference. http://bit.ly/2jjiagiI
Researchers from Singapore-MIT Alliance for Research and Technology (SMART), MIT’s research enterprise in Singapore, and National University of Singapore (NUS) have developed a unique method for generating and processing fluid droplets under previously unattainable conditions. The discovery can be transformative in a range of scientific applications including the study of biological and chemical processes, and can pave the way for more exquisite and targeted pharmaceutical and consumer products.
The new process is explained in a paper titled “Embedded droplet printing in yield-stress fluids”, published in the prestigious journal, Proceedings of the National Academy of Sciences of the United States of America (PNAS). The project is part of the National Research Foundation’s (NRF) Intra-CREATE Collaborative Grant, which enabled the collaboration between researchers from the Campus for Research Excellence and Technological Enterprise (CREATE) partner institutions SMART and NUS.
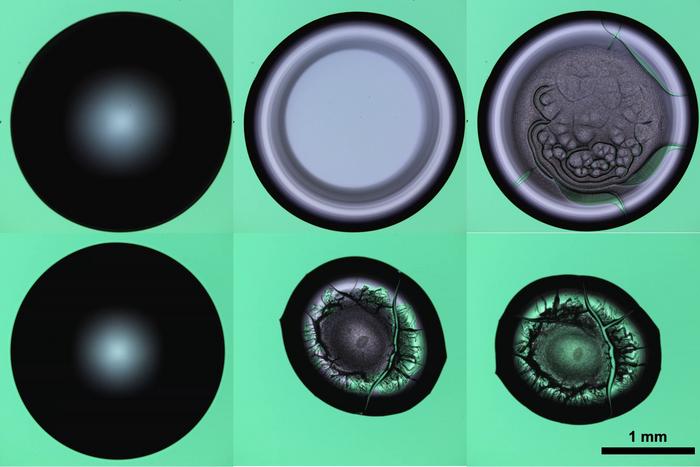
Dr Arif Zainuddin Nelson, a researcher under SMART and Intra-CREATE’s project “Advanced Manufacturing of Pharmaceutical Drug Products using Modular Microfluidic Processes”, led the development of the new method, which is the first of its kind to take advantage of yield-stress fluids to create the ideal conditions for experimentation, processing or observation of various samples. Using the embedded droplet printing approach, the research team was able to produce suspended and perfectly spherical drug-laden particles. The new approach avoids malformations that are common in conventional methods, which produce particles that are ovoid in shape and result in poor flowability during manufacturing of medicines.
“We have developed a set of tools that allows us to observe and process many different applications under this unique method, including chemical and biological reactions,” said NUS Professor Saif Khan, who is also part of the research team. “Pharmaceuticals is just one of the areas where this could produce transformative results, which is where our work is focused. We could change the way drugs are made, formulate them in a way that improves quality, revolutionise the way existing drugs are taken by patients, and envision entirely new drugs that cannot be made today.”
The embedded droplet printing method, which can also be used to alter the size and dosage of existing drugs, would be particularly useful for designing high potency medicine that needs to be taken in very small doses, such as drugs taken by cancer patients. It can also lead to more tailored medicine as the new process would make it easier to develop small batches of specialised drugs for specific patients.
“With the exception of going into space to be in zero-gravity, this method is the only way to achieve an environment where various processes can be observed in such an isolated state,” said Dr Nelson. “However, achieving a zero-gravity state is prohibitively expensive, and we have created a substantially easier and cheaper process to achieve a unique environment where chemical and biological processes are undisturbed by the outside forces.”
For pharmaceuticals, Intra-CREATE’s new microfluidic process would allow the capital costs for the formation of high-quality drugs to be circumvented, leading to potentially cheaper medication as well. The microfluidic process can also enable a range of other applications outside of the manufacturing of medicine, including:
Antibiotic testing: bacteria colonies can be cultured within each individual droplet. Different antibiotics and dosages can be tested on each droplet to quickly provide doctors and researchers with a view on potential antibiotics and cures. The unique environment allows manipulation of droplets in a way that could simulate infections
Embedded chemical reaction chambers: Microfluidic systems are able to handle a high throughput of small and precise volumes of reagents. The new process enables an improved environment for chemical reactions by removing solid boundaries, and can be used for nanoparticle production
Co-author of the research paper and Principal Investigator for SMART’s Interdisciplinary Research Group, Critical Analytics for Manufacturing Personalised-Medicine (CAMP), MIT Professor Patrick Doyle said, “The new microfluidic process can be a gamechanger in a range of scientific experimentation, and the generality and wide impact of this method couldn’t have been achieved without SMART and NUS working together.”
IMAGE SOURCE: Creative Commons




Leave a Reply