A new antibiotic created by Harvard researchers overcomes antimicrobial resistance mechanisms that have rendered many modern drugs ineffective and are driving a global public health crisis.
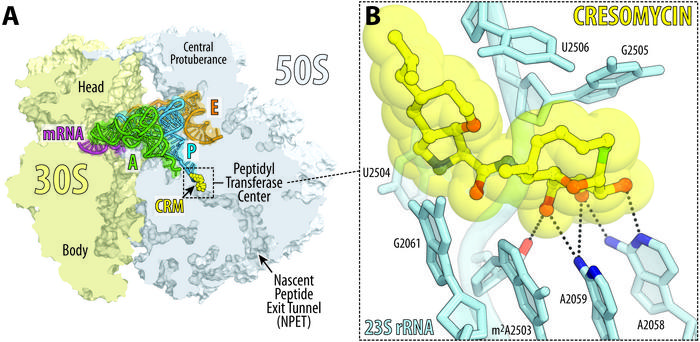
A team led by Andrew Myers, Amory Houghton Professor of Chemistry and Chemical Biology, reports in Science that their synthetic compound, cresomycin, kills many strains of drug-resistant bacteria, including Staphylococcus aureus and Pseudomonas aeruginosa.

“While we don’t yet know whether cresomycin and drugs like it are safe and effective in humans, our results show significantly improved inhibitory activity against a long list of pathogenic bacterial strains that kill more than a million people every year, compared with clinically approved antibiotics,” Myers said.
The new molecule demonstrates an improved ability to bind to bacterial ribosomes, which are biomolecular machines that control protein synthesis. Disrupting ribosomal function is a hallmark of many existing antibiotics, but some bacteria have evolved shielding mechanisms that prevent legacy drugs from working.
Sign up for the Daily Dose Newsletter and get every morning’s best science news from around the web delivered straight to your inbox? It’s easy like Sunday morning.
Cresomycin is one of several promising compounds that Myers’ team has developed, with the goal of helping win the war against superbugs. They’ll continue advancing these compounds through preclinical profiling studies, supported by a $1.2 million grant from Combating Antibiotic-Resistant Bacteria Biopharmaceutical Accelerator (CARB-X). A Boston University-based global nonprofit partnership, CARB-X is dedicated to supporting early-stage antibacterial research and development.
The Harvard team’s new molecule draws inspiration from the chemical structures of lincosamides, a class of antibiotics that includes the commonly prescribed clindamycin. Like many antibiotics, clindamycin is made via semisynthesis, in which complex products isolated from nature are modified directly for drug applications. The new Harvard compound, however, is fully synthetic and features chemical modifications that cannot be accessed through existing means.
“The bacterial ribosome is nature’s preferred target for antibacterial agents, and these agents are the source of inspiration for our program,” said co-author Ben Tresco, a Kenneth C. Griffin Graduate School of Arts and Sciences student. “By leveraging the power of organic synthesis, we are limited almost only by our imagination when designing new antibiotics.”
Bacteria can develop resistance to ribosome-targeting antibiotic drugs by expressing genes that produce enzymes called ribosomal RNA methyltransferases. These enzymes box out the drug components that are designed to latch onto and disrupt the ribosome, ultimately blocking the drug’s activity.
To get around this problem, Myers and team engineered their compound into a rigidified shape that closely resembles its binding target, giving it a stronger grip on the ribosome. The researchers call their drug “pre-organized” for ribosomal binding because it doesn’t need to expend as much energy conforming to its target as existing drugs must do.
The researchers arrived at cresomycin using what they call component-based synthesis, a method pioneered by the Myers lab that involves building large molecular components of equal complexity and bringing them together at late stages – like pre-building sections of a complicated LEGO set before assembling them. This modular, completely synthetic system allows them to make and test not just one, but hundreds of target molecules, greatly speeding up the drug discovery process.
The stakes are clear. “Antibiotics form the foundation on which modern medicine is built,” said co-author and graduate student Kelvin Wu. “Without antibiotics, many cutting-edge medical procedures like surgeries, cancer treatments, and organ transplants, cannot be done.”
Myers’ component-based synthesis research received early support from Harvard’s Blavatnik Biomedical Accelerator, part of the Office of Technology Development, which awarded funding to Myers’ lab in 2013 to enable testing of drug compounds. The Office of Technology Development protected the Myers Research Group’s innovations and, along with the Blavatnik Biomedical Accelerator, will support the research team for the duration of the CARB-X agreement. The newly awarded CARB-X funding allows the researchers to continue profiling and optimizing drug leads.
“Funding and other support from groups like the Blavatnik Biomedical Accelerator and CARB-X are essential for the discovery and development of new antibiotics,” said Curtis Keith, the Harvard accelerator’s chief scientific officer. “These innovations from the Myers Research Group have the potential to yield new drugs that will one day meet a global health need.”
IMAGE CREDIT: Yury Polikanov/University of Illinois Chicago
If you enjoy the content we create and would like to support us, please consider becoming a patron on Patreon! By joining our community, you’ll gain access to exclusive perks such as early access to our latest content, behind-the-scenes updates, and the ability to submit questions and suggest topics for us to cover. Your support will enable us to continue creating high-quality content and reach a wider audience.
Join us on Patreon today and let’s work together to create more amazing content! https://www.patreon.com/ScientificInquirer



Leave a Reply