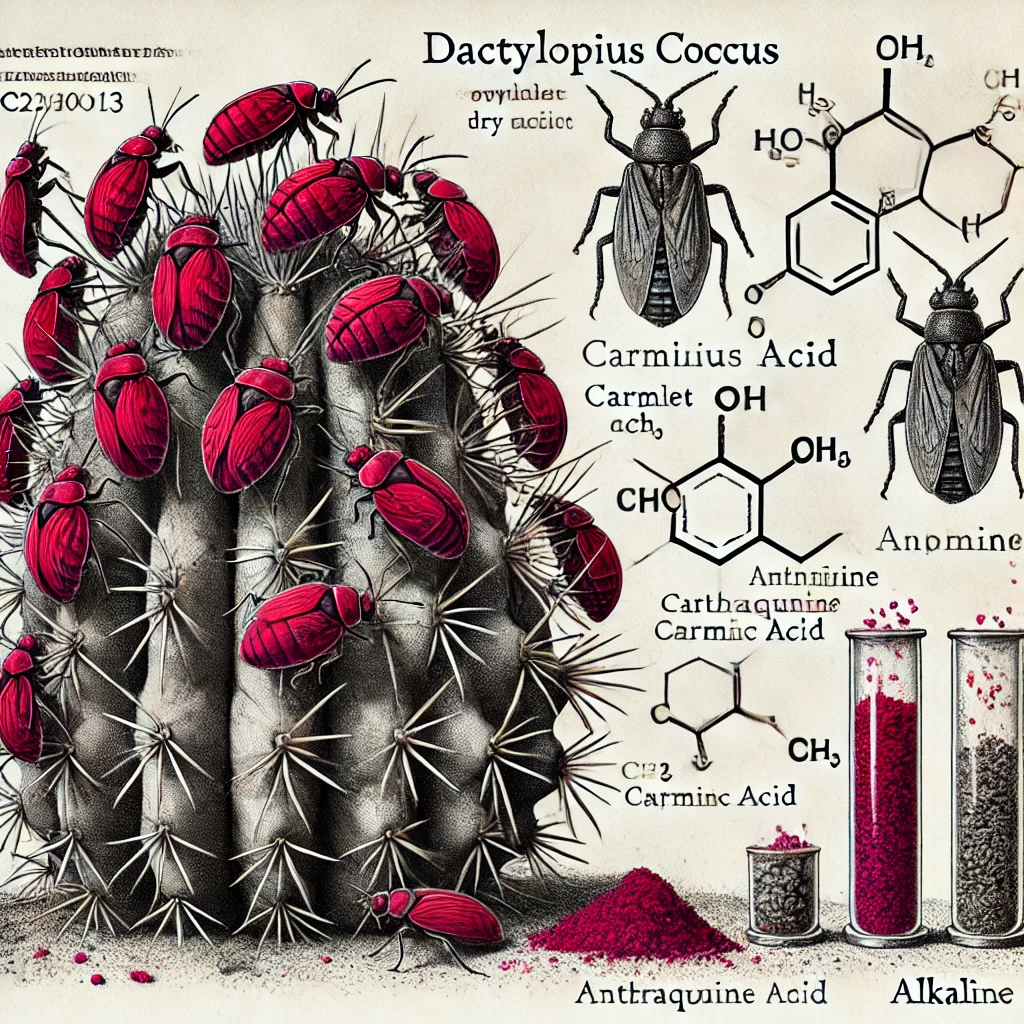
The creation of cochineal, one of history’s most prized red pigments, represents a fascinating intersection of entomology, chemistry, and human ingenuity. At its core, the science of cochineal revolves around a remarkable compound called carminic acid (C22H20O13), which constitutes approximately 20% of the female cochineal insect’s dry weight.
The process begins with the Dactylopius coccus, a scale insect that feeds exclusively on Opuntia cacti. These tiny insects produce carminic acid as a natural defense mechanism against predators. The female insects, which lack wings and remain stationary throughout most of their lives, concentrate this compound in their bodies, creating a biological factory for one of nature’s most stable red pigments
The chemistry of cochineal becomes particularly interesting when examining how it interacts with different chemical environments. In its pure form, carminic acid appears as a dark purple powder, but its color-shifting properties emerge when exposed to varying pH levels. In acidic conditions, cochineal displays brilliant scarlet hues, while alkaline environments push the color toward purple tones. This characteristic allows for a remarkable range of color possibilities through careful manipulation of chemical conditions.
The molecular structure of carminic acid explains its exceptional stability and lightfastness. The compound belongs to the anthraquinone family, characterized by a complex ring structure that creates strong chemical bonds resistant to degradation from light and heat. This stability, combined with its intense coloring power, makes cochineal particularly valuable in both historical and modern applications.
The extraction process involves precise scientific methodology. After harvesting, the insects are dried and ground into a fine powder. The carminic acid is then extracted using various solvents, with water being the traditional medium. Modern techniques may employ alcohol or other solvents to achieve different levels of purity and concentration. The extracted dye can be further processed to create carmine, a bright red aluminum lake pigment formed by precipitating carminic acid with aluminum salts.
Remarkably, cochineal’s efficiency as a colorant is extraordinary – it takes only about 2.1% cochineal by weight to create intense red hues in products, making it one of the most potent natural colorants known to science. This efficiency, combined with its safety profile and stability, ensures cochineal’s continued relevance in modern applications, from food coloring to cosmetics and textile dyes.

Titian, one of the Venetian Renaissance’s most celebrated painters, was among the many artists who harnessed the vibrant power of cochineal red to enhance his works.
Known for his masterful use of color, Titian used the deep, warm reds from cochineal pigments to enrich the skin tones, garments, and drapery in his portraits and religious paintings. This unique red not only added intensity to his compositions but also symbolized luxury and power, making it an ideal choice for portraying nobility and mythological subjects.
The cochineal pigment’s stability and brilliance allowed Titian to achieve a depth and warmth in his works that was rare for his time, helping to establish his reputation as a colorist and set a high standard for later artists in the Renaissance and beyond.




Leave a Reply