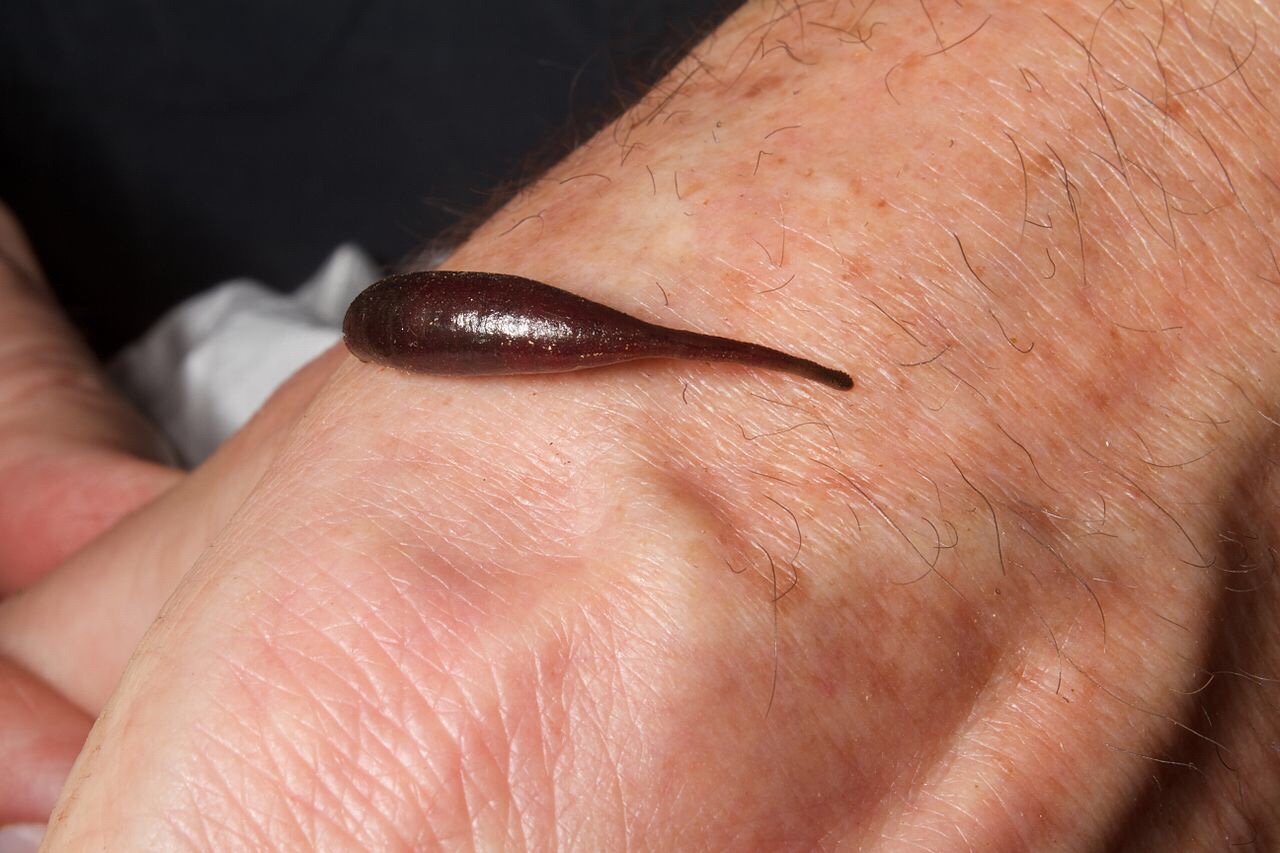
An international team of researchers recently took a deep dive into the microbiome of blood-sucking medicinal leeches and made a surprising observation: low levels of antibiotics in the animal’s environment improved the survival of antibiotic-resistant bacteria in its gut. Those resistant bacteria, in turn, displaced healthy bacteria. The findings, published this week in mBio, an open-access journal of the American Society for Microbiology, could help explain why antibiotic resistant infections have been found in patients who undergo medicinal leech therapy.
In addition, “it suggests that contamination with very low levels of antibiotics in other environments can lead to the increase in resistant bacteria,” says microbiologist Joerg Graf at the University of Connecticut, in Storrs, who led the study.
Microbiologists have long known that the overuse of antibiotics in people and animals leads to antibiotic resistance or the proliferation of germs that don’t respond to usual treatments. Antibiotic resistance can develop in the environment, too, as hospitals and pharmaceutical companies create favorable conditions for resistance by discharging large quantities of medications.
But what concentration of antibiotic exposures boost the growth of resistant microbes in the wild? The new study suggests the threshold is low. The researchers found resistant bacteria thriving in leeches exposed to less than four-hundredths of a milligram, per milliliter, of ciprofloxacin in the environment. That level represents less than 1 percent of the “clinical resistance breakpoint,” or concentration in the gut that selects for resistance.
The medicinal leech, or Hirudo verbana, is a blood-sucking, wormlike freshwater animal that secretes anticoagulants and vasodilators. Bloodletting with leeches dates back at least to the Bronze Age. In the United States, leeches are FDA-approved for tissue reconstructive surgery, which carries a high risk of blood clots. Leech therapy carries a risk of infection by Aeromonas bacteria that dwell in the leech gut, so patients are given the antibiotic ciprofloxacin as a preventive measure. However, the last few years have brought an uptick in the number of patients who develop ciprofloxacin-resistant Aeromonas infections.
The new study may help explain why, says Graf, whose lab focuses on understanding interactions between beneficial bacteria and their animal host. Previous studies on leech gut bacteria have been based on biochemical observations in the lab, but Graf’s group’s findings are the first to use genomic data to link the animal source of the infections – the leech’s gut – to the patients themselves. Using genomic sequencing, the researchers determined that some clinical resistant strains and isolates from the leech guts shared identical sequences for 16 housekeeping genes, demonstrating the link between the two at a molecular level.
“We showed how closely related the strains are,” says Graf.
The researchers also observed that adding very small amounts of ciprofloxacin to blood meal eaten by leeches provided a growth advantage to resistant strains. The study suggests that the leeches used for medicinal purposes have been exposed at some point to powerful antibiotics, which allowed the ciprofloxacin-resistant strains to proliferate. However, he says the source of that original exposure remains an open question. (Other researchers have theorized that the antibiotic might have been present.
IMAGE SOURCE: Creative Commons
The Scientific Inquirer needs your support. Please visit our Patreon page and discover ways that you can make a difference. http://bit.ly/2jjiagi


Leave a Reply