Genesis Mission: Grand AI Vision, Hollow Science Foundation
Donald Trump’s “Genesis Mission” executive order frames AI as a moonshot-scale catalyst for scientific breakthroughs, proposing a massive integrated platform trained on federal datasets to accelerate discovery. Critics argue the initiative is untethered from reality. The order is vague on funding, partnerships, and governance, arriving after deep cuts to research agencies, disrupted grants, workforce instability, and damage to public datasets. Scientists warn that AI cannot compensate for eroded institutions, missing data, and shrinking talent pipelines. Former science officials say rebuilding trust, restoring funding, stabilizing labs, and fixing visa pathways are prerequisites for success. The mission’s aggressive timelines, selective focus on politically favored fields, and industry-leaning leadership raise concerns about politicized science and overpromised AI capabilities. Without repairing the underlying research ecosystem, critics say Genesis risks becoming hype rather than progress. (Ars Technica)
Epigenetic clocks go head-to-head across 174 diseases
Epigenetic “clocks” promise to estimate biological age, but most studies test only a few clocks in small samples. A new open-access Nature Communications analysis runs a large, apples-to-apples comparison: 14 widely used clocks evaluated in 18,859 people, tested against 174 incident disease outcomes plus all-cause mortality over 10 years of follow-up. The authors report that “second-generation” clocks—those trained to capture health risk rather than just chronological age—perform better at predicting who goes on to develop disease and who dies, with particularly strong signals for respiratory and liver-related outcomes. The paper is a practical field guide for anyone using DNA-methylation aging measures: it helps researchers pick clocks that match their question (risk prediction vs. age tracking) and highlights how much results can change depending on the clock you choose. (Nature)
A non-invasive route to “skin age” via epigenetic predictors
Aging biomarkers are increasingly used in trials of lifestyle, cosmetics, and therapeutics—but repeatedly sampling internal tissues is impractical. This open-access npj Aging article introduces epigenetic age predictors designed for non-invasive assessment of human skin, aiming to estimate biological aging signals from skin-derived methylation data rather than relying on blood alone. By focusing on skin specifically, the work targets a major gap in “organ-specific” aging measurement: the skin ages under unique exposures (UV, pollutants, barrier stress) and is often the first tissue where people seek measurable “rejuvenation.” The study’s contribution is methodological and translational: tools meant to make skin-aging readouts more feasible for longitudinal monitoring and intervention studies, including those that want tissue-relevant endpoints rather than systemic averages. (Nature)
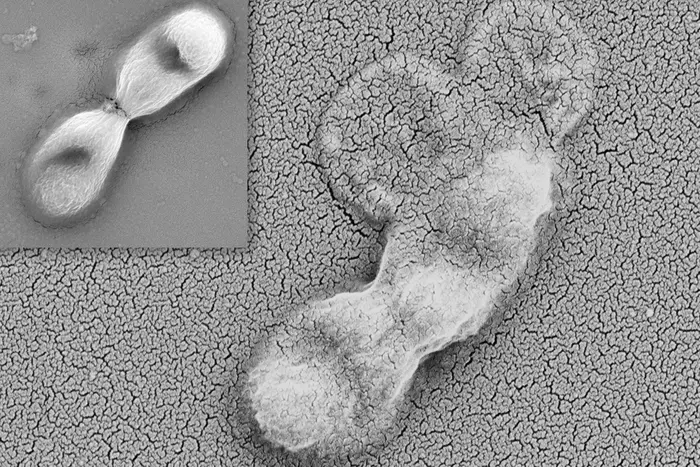
Mitochondrial supercomplex booster extends mouse lifespan and healthspan
A Japanese team reports that a mitochondrial protein involved in assembling respiratory “supercomplexes” can meaningfully shift aging biology in mice. Using COX7RP-transgenic animals with higher lifelong expression of the protein, the researchers observed a ~6.6% increase in average lifespan versus wild-type controls, alongside multiple healthspan-linked improvements: better glucose homeostasis via improved insulin sensitivity, healthier lipid profiles (lower triglycerides and total cholesterol), enhanced muscle endurance, and reduced liver fat accumulation. Mechanistically, tissues showed increased respiratory supercomplex formation and higher ATP production; white adipose tissue also showed higher NAD+, lower reactive oxygen species, reduced β-galactosidase (a senescence marker), and lower expression of inflammatory/SASP-associated genes by single-nucleus RNA-seq in older mice. The implication: targeting supercomplex assembly may be a viable longevity strategy. (EurekAlert!)
Dark chocolate’s theobromine linked to “younger” biological age signals
A King’s College London-led study reports an association between circulating theobromine—a cocoa alkaloid abundant in dark chocolate—and lower biological age estimates in humans. The analysis compared blood levels of theobromine with blood-based biological aging markers, including DNA methylation–based measures (epigenetic “bookmarks” that shift with age) and telomere length (with shorter telomeres generally associated with aging and age-related disease). Across two European cohorts (TwinsUK, n=509; and KORA, n=1,160), people with higher theobromine levels tended to show biological ages lower than their chronological ages. The researchers tested other cocoa/coffee metabolites but report the signal appeared specific to theobromine. They explicitly caution this does not mean “eat more chocolate,” since chocolate also carries sugar and fat; the near-term value is as a clue for diet–epigenome mechanisms worth probing experimentally. (EurekAlert!)
Machine learning spots “slow-aging” signatures in mouse blood—before lifespan data arrives
Lifespan studies in mice can take three years, slowing the hunt for geroprotective drugs. This open-access GeroScience paper proposes a shortcut: plasma metabolomic (≈12,000 features) and proteomic/peptidomic (≈17,000 features) signatures that distinguish normal from “slow-aging” mice exposed to known longevity interventions. Using XGBoost regression across 278 mice and five anti-aging interventions, models trained on four interventions could still predict significantly higher lifespan extension in the omitted (unseen) intervention group—suggesting the signatures generalize beyond a single drug. The models also separated long-lived mutant mice from controls and even transferred across sex (male-trained models discriminating treated vs. control females). A standout signal class: triglycerides, where longer-chain species tended to be higher in slow-aging mice while shorter-chain species decreased. The work frames blood “omics” as a triage tool for prioritizing candidate longevity therapies. (Springer)

Genetic analysis links multimorbidity risk with biological aging pathways
As people live longer, the central clinical challenge becomes multimorbidity—accumulating multiple chronic conditions rather than a single diagnosis. A new GeroScience paper reports a “large genetic study” that examines links between multimorbidity and biological aging, aiming to identify shared genetic architecture that could explain why certain individuals experience clustered diseases as they age. The work sits at a key intersection for longevity medicine: if multimorbidity and aging share underlying genetic drivers, then interventions targeting those shared pathways might compress morbidity rather than just extend lifespan. While the paper is genetics-forward (rather than an intervention trial), it is directly relevant to risk stratification—who is predisposed to age into multiple conditions—and to the search for targets that are upstream of many diseases at once. (Springer)
Aptamers tag senescent “zombie” cells—opening a path to smarter senolytics
Senescent cells contribute to aging and diseases ranging from cancer to Alzheimer’s, but identifying them in living tissue without disrupting nearby cells remains a major bottleneck. Mayo Clinic researchers, writing in Aging Cell, report an aptamer-based strategy: short synthetic DNA sequences that fold into 3D shapes and bind target proteins on cell surfaces. Starting from an enormous pool (over 100 trillion random sequences), the team selected rare aptamers that recognize surface proteins enriched on senescent cells and can label them in mouse-cell experiments. A key advantage is discovery: because there is no universal senescence marker, the authors let aptamers “choose” binding targets rather than prespecifying one protein. If the approach translates to human cells, it could improve senescence detection for imaging, tissue profiling, and—critically—more selective senolytic therapies that hit senescent cells while sparing healthy neighbors. (ScienceDaily)
Lifestyle factors tied to “brain age” differences of up to 8 years
A University of Florida team used MRI scans plus machine learning to estimate “brain age” and track how it changes—particularly in adults with or at risk of knee osteoarthritis and chronic musculoskeletal pain. Over two years, the study followed 128 midlife and older adults, comparing each person’s brain-age estimate to their chronological age (the “brain age gap”). Certain stressors (including chronic pain and socioeconomic disadvantage) were associated with older-appearing brains, but the more durable signal in this dataset came from protective behaviors and psychosocial factors: restorative sleep, stress management, maintaining supportive relationships, avoiding tobacco, and healthy body weight. Participants reporting the most protective factors started with brains that looked about eight years younger than expected and showed slower brain aging over follow-up. The takeaway for longevity is not a miracle lever, but an additive model: each incremental healthy factor corresponded to measurable neurobiological benefit in this imaging-based metric. (ScienceDaily)
Why metabolism–inflammation “crosstalk” is a longevity control panel
This Frontiers in Endocrinology editorial synthesizes a core theme in aging biology: metabolism and inflammation don’t run as separate systems—they amplify each other over time, shaping senescence, tissue dysfunction, and disease vulnerability. The piece frames mitochondria, NAD+ metabolism, oxidative stress signaling, immune–metabolic rewiring, and senescence-associated inflammatory programs (including SASP) as interconnected levers that can push organisms toward either resilience or decline. Rather than presenting a single new dataset, the editorial’s value is directional: it maps a “research topic” landscape, highlights recurring mechanistic motifs (mitochondrial stress, immunometabolism, redox imbalance, cell death pathways), and points to gaps where the field still lacks causal clarity or actionable biomarkers. For longevity readers, it’s a useful way to contextualize why interventions as different as exercise, diet composition, NAD+ precursors, senolytics, and mitochondrial modulators often converge on overlapping downstream signatures—because the metabolism–inflammation loop is a shared highway for many age-related phenotypes. Published December 15, 2025. (Frontiers)
Intrinsic capacity tracked across three cohorts to study cardiometabolic multimorbidity
A large Frontiers in Endocrinology research article uses the World Health Organization’s “intrinsic capacity” (IC) framework—cognition, psychological health, sensory function (vision/hearing), vitality, and locomotion—as a structured way to quantify functional reserve in older adults. The authors harmonize IC measures across three major prospective cohorts: CHARLS (China), ELSA (England), and HRS (United States). After exclusions for missing data and baseline multimorbidity, the final analytic sample reported on the page is 11,916 participants over age 50. The study then evaluates IC at baseline and follow-up (including cumulative IC scores and IC trajectory categories such as decline, improvement, and persistent impairment) in relation to cardiometabolic multimorbidity outcomes. The key contribution is scale and comparability: it treats “aging resilience” as a measurable construct that can be tracked over time and tested against hard clinical clustering, rather than relying on single-disease endpoints. Published December 17, 2025. (Frontiers)
Microsoft Finally Retires RC4, Closing a Longstanding Security Backdoor
Microsoft is deprecating RC4, an obsolete encryption cipher supported by Windows for 26 years, after years of exploits and mounting criticism. Originally embedded in Active Directory in 2000, RC4 was known to be weak almost immediately after its algorithm leaked in the 1990s, yet it persisted as a default fallback for authentication. Attackers repeatedly exploited this weakness through Kerberoasting, including in the 2024 Ascension healthcare breach that disrupted 140 hospitals and exposed millions of records. By mid-2026, Windows domain controllers will disable RC4 by default and rely solely on AES-SHA1, a far more secure alternative already widely supported. Microsoft is rolling out logging tools and PowerShell scripts to help administrators identify lingering RC4 dependencies, particularly in legacy systems that may still rely on it. (Ars Technica)




Leave a Reply