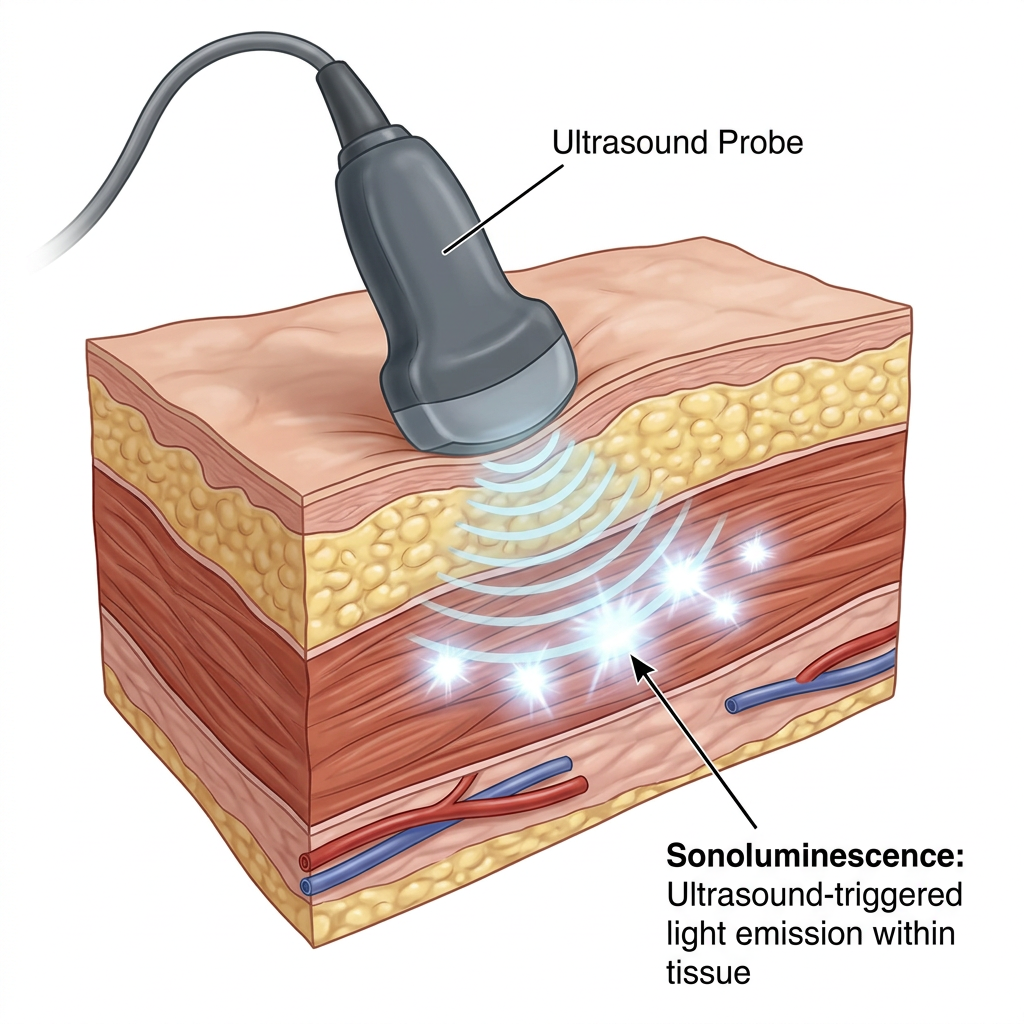
NHS TRIAL TESTS ULTRASOUND BRAIN DEVICE FOR MOOD DISORDERS
A groundbreaking NHS trial will test a brain-computer interface (BCI) that uses ultrasound to alter brain activity and improve patients’ mood. The device, implanted beneath the skull but outside the brain, sends targeted pulses to stimulate neurons, with a focus on treating conditions like depression, addiction, OCD, and epilepsy. The £6.5m trial, involving around 30 patients, is funded by the UK’s Advanced Research and Invention Agency (Aria). Unlike traditional invasive BCIs, this device uses ultrasound to modify activity across multiple brain regions. The trial aims to offer a less invasive, safer alternative for conditions that resist current treatments. While the technology holds promise, ethical concerns about data privacy, brain enhancement, and neuro-discrimination persist. The trial will also address safety considerations, such as minimizing tissue heating, and will run for three and a half years, potentially paving the way for broader clinical applications. (The Guardian)
TIKTOK RESUMES U.S. OPERATIONS AMIDST EXECUTIVE ORDER DELAY
TikTok resumed operations in the U.S. on Sunday after President-elect Donald Trump announced plans to issue an executive order delaying a federal ban on the app. The ban, set to take effect due to a 2024 law, requires app stores and cloud providers to stop distributing TikTok unless sold by its Chinese parent company, ByteDance. This law raised concerns about national security and the app’s potential use for gathering data or spreading propaganda. Trump’s executive order aims to extend the deadline, allowing for negotiations to protect U.S. interests. The order could challenge the rule of law, as it would temporarily override a law upheld by the Supreme Court. TikTok, which claims 170 million U.S. users, celebrated its return, but legal experts caution that the ban remains enforceable. The situation raises questions about presidential authority, with potential legal challenges ahead. Lawmakers from both parties continue to debate the future of TikTok. (New York Times)
TRUMP REVERSES TIKTOK BAN DECISION AHEAD OF RETURN
During his first term, Donald Trump led the effort to ban TikTok, citing national security concerns. However, on the eve of his return to the White House, he reversed his position, announcing plans to delay the ban with an executive order aimed at protecting national security. This move allowed TikTok to resume operations, much to the relief of its 170 million U.S. users. The law, passed with bipartisan support, required TikTok’s parent company, ByteDance, to sell its U.S. operations by Sunday. Although Trump’s legal authority to override the law is unclear, his actions signal a shift in the debate over technology and national security. TikTok’s CEO expressed gratitude for Trump’s support, while legal experts warn the executive order could face challenges. Trump’s efforts reflect his evolving stance on social media, which played a significant role in his campaign, particularly with young voters. (AP)
ASPR AWARDS $375 MILLION FOR COVID-19 DRUG DEVELOPMENT
The Administration for Strategic Preparedness and Response (ASPR) announced a $375 million grant to Shionogi, Inc. to develop a drug, S-892216, to prevent COVID-19 in immune-compromised individuals. This long-acting protease inhibitor blocks the SARS-CoV-2 virus from replicating in human cells and is intended for pre-exposure prophylaxis in people who may not respond well to vaccines. The funding comes from Project NextGen, a $5 billion initiative to advance COVID-19 therapeutics. Shionogi plans to submit an Investigational New Drug application and begin phase 1 clinical trials this year. Additionally, CARB-X awarded $610,000 to Justus Liebig University Giessen in Germany to develop a novel peptide antibiotic targeting the BamA protein in gram-negative bacteria. This antibiotic could offer broad-spectrum activity and help combat antibiotic-resistant pathogens. With this funding, CARB-X has supported 108 early-stage projects focused on antibiotic resistance. (CIDRAP)

STUDY SHOWS RNA MOLECULE COULD REVERSE SIGNS OF AGING
A recent study published in *Cell Metabolism* suggests that injecting old mice with an RNA molecule, miR-302b, could reverse some signs of aging, including extended lifespan, hair regrowth, and improved physical and mental abilities. The treatment targets cellular senescence, a key feature of aging where cells lose their ability to replicate, triggering inflammation and contributing to diseases like cancer, cardiovascular issues, and cognitive decline. Researchers used exosomes containing miR-302b, a molecule involved in gene regulation, to rejuvenate senescent cells in mice aged 60-70 in human years. Mice treated with miR-302b lived an average of 4.5 months longer, exhibited improved physical abilities, and showed reduced inflammation and better cognitive performance. While promising, experts caution that more research is needed to confirm the effects and determine whether they can safely translate to humans, as prolonged treatments could potentially lead to adverse effects such as cancer development. (Nature)
JAPAN APPROVES TECOVIRIMAT FOR MPOX DESPITE INEFFECTIVE STUDIES
Japan’s regulatory agency approved the antiviral drug tecovirimat (TPOXX) for treating mpox, smallpox, and cowpox, despite recent studies showing it ineffective for mpox. Tecovirimat, which works by blocking virus replication, was initially considered promising, with prior trials showing success in monkeys. It was approved by the EU and UK in 2022 for mpox, but two recent clinical trials, PALM007 and STOMP, found no benefit for humans infected with mpox. In these studies, tecovirimat did not speed healing or reduce mortality. The approval by Japan’s Pharmaceuticals and Medical Devices Agency (PMDA) raised concerns due to the lack of efficacy data and its reliance on safety trials with healthy volunteers. Experts question the basis for Japan’s decision, as it appears to conflict with negative study results. The approval could lead to reconsiderations by other regulators, including the FDA, which has approved tecovirimat for smallpox based on animal studies but is now facing pressure to reassess its efficacy. (Science)
AI SIMULATES ALTERNATIVE EVOLUTIONARY PATHWAYS IN PROTEIN DESIGN
Scientists are exploring whether evolution could have unfolded differently, with some arguing that human existence might not have been inevitable. A new study, published in *Science*, uses artificial intelligence (AI) to simulate alternative evolutionary pathways in protein design, revealing potential evolutionary routes nature did not take. The AI system, EvolutionaryScale’s Model 3 (ESM3), functions similarly to generative language models like ChatGPT, but instead of text, it generates proteins. Trained on vast biological data, ESM3 created a new green fluorescent protein (esmGFP) that is only 58% similar to existing versions, simulating 500 million years of evolution. This discovery suggests that nature could have followed different paths in protein evolution. Researchers argue this supports the theory of contingency in evolution, as proposed by Stephen Jay Gould, showing that small changes could lead to drastically different outcomes. AI is now seen as a powerful tool to explore biological possibilities and understand evolutionary processes. (El Pais)
Thanks for reading. Let’s be careful out there.
WORDS: The Biology Guy.




Leave a Reply