In their groundbreaking research into heart calcification, scientists Bruce Fouke and Mayandi Sivaguru explore the complex processes behind this common yet often fatal condition. With backgrounds in geology and biology, respectively, Fouke and Sivaguru have spent over 15 years merging their expertise to investigate the formation and progression of calcification within the human cardiovascular system. Their collaborative approach has led to significant insights, particularly in distinguishing the roles of amorphous calcium phosphate (ACP) and hydroxyapatite (HAP) in heart valve calcification. Their findings offer hope for developing new strategies to slow down or even reverse the calcification process, potentially leading to less invasive treatments for those affected.
In this interview, they discuss the prevalence of calcification, the dynamics of mineral transformation in the heart, and the future clinical applications of their research.

Just starting with a simple question, how common is calcification?
Bruce Fouke: Shiv, you wanna kick off here?
Mayandi Sivaguru: Oh, yes, sure. So, the number is around 18 million people affected.
Bruce Fouke: That’s for heart disease in total.
Mayandi Sivaguru: Yes, in total.
Bruce Fouke: Total heart disease. Of those, we don’t know the exact number. It could be as many as half or more. For people 65 and older, it’s the number one cause of heart disease.
Mayandi Sivaguru: Yes, the incidence and calcification levels increase with age. Based on our estimates, even within the United States, several hundred thousand people are affected every year. It’s a fatal end-stage calcification.
Unfortunately, it takes a toll on human lives. That’s just in the United States, but worldwide, the number we cited in our paper is 18 million. Some people survive for a long time with it, but others do not. It depends on the rate of calcification.
I have a close friend with calcification, and she still lives. It’s not an immediate death sentence; it’s progressive. When it reaches the point that the valve is no longer able to function, that’s when it becomes critical.
In my case, my mother-in-law experienced this. The only solution so far is to replace the aortic valve with either a synthetic valve or one isolated from a pig. This can allow the person to survive for another 10 to 15 years, but those valves also get calcified after the surgery. It’s not a complete solution. When patients complain about shortness of breath, doctors perform an angiogram to determine the next steps. There are no definitive treatments.

Is calcification most common in terms of valves?
Mayandi Sivaguru: It is everywhere in the vasculature, including coronary arteries and multiple heart valves. All of these places can get calcification, not just the valves.
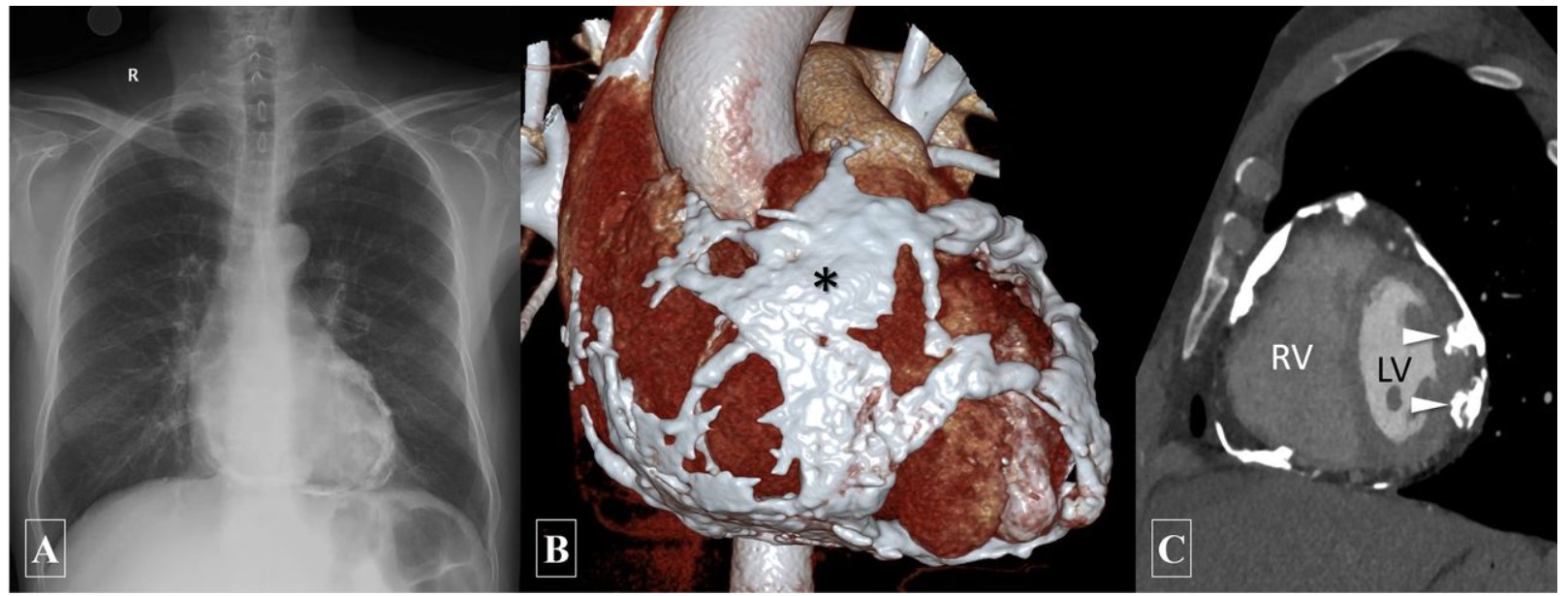
Bruce Fouke: And you know, Marc, we’ve studied this with our colleagues at UCLA Health in Los Angeles, which is a primary center for cardiovascular calcification surgeries. We were very fortunate to meet and collaborate with them. They have a special donor program called Legacy One, from which we received cadaver heart samples.
Using micro-CT (microcomputed tomography) here, Shiv ran the hearts, and we found that calcification occurs in the aortic valves, coronary arteries, and many other parts of the cardiovascular system. It can even extend to the legs and arms, affecting the entire cardiovascular system.
Can you explain in layman’s terms what amorphous calcium phosphate (ACP) and hydroxyapatite (HAP) are, and their roles in valve calcification?
Bruce Fouke: Sure, Marc. Shiv and I have worked together for 15 years on this. My background is in geology, and Shiv’s is in biology, but we’ve essentially switched roles over time. This gives us a unique perspective.
There are two important aspects of minerals in nature: chemical composition and mineralogy. Chemical composition is what elements make up the mineral, and mineralogy is the geometric arrangement of those elements.
Mineral deposits can have two large-scale formations. One is amorphous, meaning the chemical ions are not well-structured or ordered. The other is crystalline, where the elements are arranged in a geometric pattern.
Amorphous calcium phosphate (ACP) is not rigidly crystalline and can move quickly from one chemical composition to another. We call it shape-shifting. In contrast, hydroxyapatite (HAP) is a stable, crystalline form.
Previously, ACP had not been well-identified in aortic valve leaflets, with only about four papers suggesting its presence. Most research teams assumed it was HAP because HAP is the mineral of bone. This assumption was logical given our body’s composition and the nature of bone.
The distinction between ACP and HAP is crucial because they have different dynamics in precipitation and behavior. ACP is involved in the earlier stages of calcification and behaves differently from HAP. Understanding the mineral and chemistry helps us understand the system better. For example, ACP behaves differently under various temperature and pH conditions than HAP and interacts differently with proteins.

That leads into my next question. How does the stabilization of osteopontin and collagen containment slow down the transformation of ACP into HAP during the calcification process?
Mayandi Sivaguru: Good question. To add to what Bruce explained, we published a paper titled “Lack of Order, Abundance of Possibilities.” ACP lacks a specific structure, giving it enormous possibilities in nature, from invertebrates to vertebrates, including humans.
ACP is thermodynamically unstable, while HAP is stable and seeks equilibrium. When stabilized, it forms a structure that can be lethal, like sharp kidney stones. The system tries to prevent this final stabilization step in certain locations.
Proteins and polypeptides act as inhibitors. Even magnesium can prevent ACP from transforming into other forms. ACP can convert into many other forms depending on the ionic environment and pH.
Osteopontin, a paradoxical protein, can both promote and inhibit calcification. When ACP is coated with phosphorylated osteopontin, the phosphate can bind to osteopontin instead of forming ACP, preventing further transformation. Collagen also plays a role by containing the calcified ACP and preventing exposure to external ions or liquids, like blood. This containment prevents the ACP from becoming a more crystalline and damaging form.
Bruce Fouke: To summarize, our bodies are destined to calcify tissues because blood contains calcium, phosphate, and water. However, our bodies have evolved to slow this process. Osteopontin, magnesium, and collagen are key agents in slowing down calcification. By understanding how nature slows it down, we can use similar techniques to slow it further.
Speaking of doom, how do cholesterol crystals contribute to the calcification process?
Mayandi Sivaguru: Cholesterol is another factor. Excess cholesterol, whether from diet or genetic disorders, needs to be stored in the body. It accumulates in various locations, including the heart.
In the heart, cholesterol can accumulate in the collagen region, heart tissue, leaflets, and coronary arteries. Cholesterol undergoes diagenetic transformation, where lipid molecules coalesce under the right conditions to form crystalline cholesterol, often needle-like. This can happen simultaneously with ACP formation, as shown in our paper. Cholesterol is particularly prevalent in the coronary arteries.
I have a few more questions. Your study mentions a transdisciplinary approach called Geo. Biomed. Can you elaborate on what that is and how you applied it to your research?
Bruce Fouke: Sure. Geo. Biomed is something Shiv and I developed over years of work. It’s the marriage of geology, biology, and medicine.
Before we started doing human medicine, we were focused on geobiology, studying life-mineral-water interactions in natural environments like coral reefs, oil fields, and Yellowstone’s hot springs. We call this the “eternal triangle.”
We then transitioned to kidney stones, which are similar to coral reefs and hot springs in terms of their system. After that, we moved to the heart and now to breast cancer, which also involves calcifications. Geo. Biomed combines the processes and techniques from these fields to study the human body as a product of life and earth coevolution.
This approach also includes various techniques, such as optical, laser, X-ray, and electron microscopy, as well as Raman spectroscopy and microcomputed tomography. These techniques allow us to characterize calcifications at different scales.
We have strategically invested in building Geo. Biomed laboratory facilities that allow us to analyze everything from proteins and DNA to minerals and blood flow, integrating all these aspects into medical interventions.
Your research elucidates how the calcification process occurs and how it can be slowed down. What are the potential clinical applications and implications of your findings?
Mayandi Sivaguru: The main clinical implication is that while we can’t prevent calcification, we can slow it down. Osteopontin, particularly its phosphorylated forms, can inhibit further transformation of ACP into more harmful forms. If we can increase the production of this protein in people who lack it, it could be beneficial.
Additionally, natural compounds, like plant extracts, could prevent or dissolve calcifications. Microfluidic experiments can help identify effective treatments, which can then be applied in clinical trials. Currently, the only treatment is invasive surgery to replace damaged aortic valves. Identifying calcification early and treating it could provide less invasive options.
Bruce Fouke: We’ve come up with the first systematic sequence of events that track calcification from its origin to full development. This roadmap allows us to identify multiple intervention points, either to slow it down or dissolve it. This comprehensive understanding opens up many potential treatments.
What’s next for you in terms of research in this field?
Mayandi Sivaguru: We’re currently working on breast biopsies from the Mayo Clinic to understand the sequence of events in breast calcifications. We aim to find commonalities between calcifications in the kidney, heart, and breast.
Understanding why large crystals form in the kidney but not in the heart or breast, despite similar fluidic systems, is crucial. This research will help us put together a comprehensive picture of calcification in the human body and how to slow it down or dissolve it, making life more manageable for patients.
Bruce Fouke: We call this “universal biomineralization of the human body.” Our work in the kidney, heart, and breast follows a sequence that has allowed us to see patterns and processes across different systems. This grand synthesis views the human body as a microcosm of earth processes, integrating geological, biological, and medical insights.
Sign up for the Daily Dose Newsletter and get the morning’s best science news from around the web delivered straight to your inbox? It’s easy like Sunday morning.




Leave a Reply