A growing repertoire of cell and molecule-based immunotherapies is offering patients with indomitable cancers new hope by mobilizing their immune systems against tumor cells. An emerging class of such immunotherapeutics, known as T cell bispecific antibodies (TCBs), are of growing importance with several TCBs that the U.S. Food and Drug Administration (FDA) approved for the treatment of leukemias, lymphomas, and myelomas. These antibody drugs label tumor cells with one of their ends, and attract immune cells with another end to coerce them into tumor cell killing.
One major challenge in the development of TCBs and other immunotherapy drugs is that the antigens targeted by TCBs can be present not only on tumor cells, but also healthy cells in the body. This can lead to “on-target, off-tumor” cell killing and unwanted injury of vital organs, such as the kidney, liver, and others, that can put patients participating in clinical trials at risk. Currently, there are no human in vitro models of the kidney that sufficiently recapitulate the 3D architecture, cell diversity, and functionality of organs needed to assess on-target, off-tumor effects at a preclinical stage.
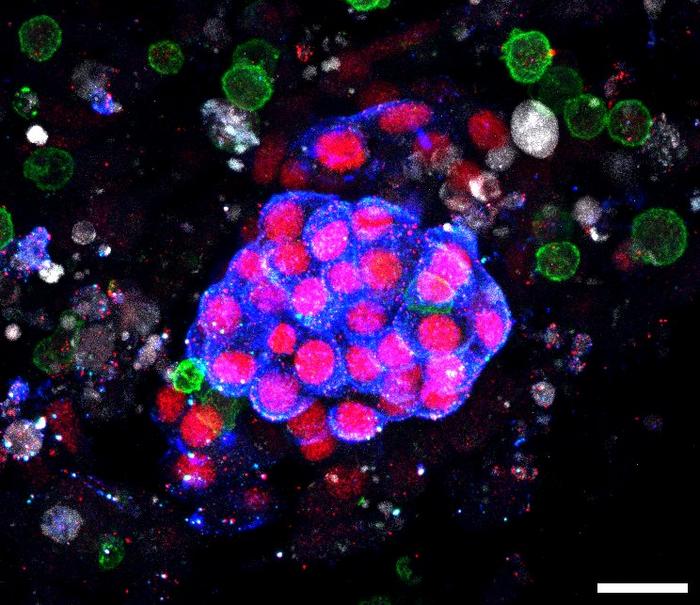
Now, a new cross-disciplinary, cross-organizational study created an immune-infiltrated kidney tissue model for investigating on-target, off-tumor effects of TCBs and potentially other immunotherapy drugs. The team of bioengineers and immune-oncologists who performed the study at the Wyss Institute for Biologically Inspired Engineering at Harvard University, Harvard John A. Paulson School of Engineering and Applied Sciences (SEAS), Harvard Medical School (HMS), and the Roche Innovation Centers in Switzerland and Germany, developed an immune-infiltrated human kidney organoid-on-chip model composed of tiny kidney tissue segments that contain vasculatureand forming nephrons, which can be infiltrated by circulating immune cells. They used this model to understand the specific toxicity of a pre-clinical TCB tool compound that targets the well-characterized tumor antigen Wilms’ Tumor 1 (WT-1) in certain tumors. Importantly, WT-1 is also expressed at much lower levels in the kidney, making it an important organ to study its potential on-target, off-tumor effects in. Their findings are published in PNAS.
If you enjoy the content we create and would like to support us, please consider becoming a patron on Patreon! By joining our community, you’ll gain access to exclusive perks such as early access to our latest content, behind-the-scenes updates, and the ability to submit questions and suggest topics for us to cover. Your support will enable us to continue creating high-quality content and reach a wider audience.
Join us on Patreon today and let’s work together to create more amazing content! https://www.patreon.com/ScientificInquirer
“Together with our collaborators at Roche, we extended our vascularized kidney organoid-on-chip model to include an immune cell population that contains cytotoxic T cells with the potential to kill not only tumor cells, but also other cells that present target antigens,” said Wyss Core Faculty member Jennifer Lewis, Sc.D., the study’s senior author. “Our pre-clinical human in vitro model provides important insights regarding which cells are targeted by a given TCB and what, if any, off-target damage arises.” Lewis is also the Hansjörg Wyss Professor of Biologically Inspired Engineering at SEAS and co-leader of the Wyss Institute’s 3D Organ Engineering Initiative.
Incorporating immunity into a kidney organoid-on-chip
In 2019, Lewis’ group, together with that of Joseph Bonventre, M.D., Ph.D. at Brigham and Women’s hospital along with co-author Ryuji Morizane, M.D., Ph.D., found that exposing kidney organoids created from human pluripotent stem cells to the constant flow of fluids during their differentiation enhanced their on-chip vascularization and maturation of glomeruli and tubular compartments, relative to static controls. The researchers’ observations were enabled by a 3D printed millifluidic chip, in which kidney organoids are subjected to nutrient and differentiation factor-laden media flowed at controlled rates during their differentiation. The chip device allows researchers to directly observe the tissue using confocal microscopy through a transparent window in real-time.
“Given that this in vitro model represents most of the cell types in the kidney and incorporates the immune system, itcould support the assessment of on and off-target effects from TCBs as well as complex cellular interactions,” said Kimberly Homan, Ph.D., a former postdoctoral researcher in Lewis’ lab, first author of the initial work, and a co-corresponding author of this new study. Homan has since left Lewis’ lab to join Genentech as Director of the Complex in vitro Systems lab where she continued to provide expertise to the project collaborators.
Approaching this challenge, the team first advanced their in vitro system to enable the continuous circulation of human immune cells, collectively known as peripheral blood mononuclear cells (PBMCs), through it and the retrieval of samples for the analysis of immune cell-secreted proteins. They demonstrated that an optimal flow rate was able to support the viability of “immune effector” cells, a subpopulation of PBMCs with the ability to kill other cells, over a 5-day period, and that these cells actually infiltrated the nephron-like structures and microvessels present in the kidney organoids on chip.
The preclinical WT-1-targeting tool TCB (WT1-TCB) was created to specifically bind to the WT-1 antigen when presented by the HLA protein on the surface of target cells, in that case WT-1-expressing tumor cells. HLA helps the immune system distinguish between proteins on healthy cells versus those produced by tumors and foreign invaders by familiarizing it with the body’s normal protein repertoire. The team first investigated whether the normal WT-1 protein was expressed in any of the key kidney cell populations. They found that WT-1 was expressed by podocytes, but was undetectable in the proximal and distal tubule cells. Moreover, they found that a significant proportion of these differentiated kidney cell types also expressed HLA.
Putting TCBs to the test
To understand specific targeting effects of WT1-TCB, the researchers compared them to those produced by a non-specific TCB that could bind antigens on all kidney cell types (ESK1-like TCB, a positive control), and a TCB that could only bind immune cells (DP47, a negative control). The three compounds indeed elicited strikingly different effects when introduced into the kidney organoid-on-chip model alongside PBMCs under high-flow conditions over five days. As expected, DP47 caused very few cells to die, while the ESK1-like TCB targeted and killed all cell types in a dose-dependent manner by recruiting immune effector cells.
“Importantly, our central finding was that the WT1-TCB tool compound resulted in the selective killing of WT-1-expressing podocytes in the kidney organoids, while it did not affect cells in the distal and proximal tubules. This compellingly demonstrates that our engineered human in vitro kidney organoid-on-chip system has utility as a preclinical drug development tool for assessing on-target, off-tumor toxicities of TCBs as a new class of immunotherapeutics,” explained Katharina Kroll, Ph.D., a co-first author of this study who performed her graduate work with Lewis and now is a Postdoctoral Fellow in her group. However, an important caveat of the current kidney organoid-on-chip system is that the TCBs and PBMCs are not delivered via the same route as they would be in vivo, where they access kidney cells through the perfusion and filtration of blood within the glomerular compartment. However, the authors hypothesize that in vivo access to the WT1 target on glomerular cells by TCBs would be minimal. To test this hypothesis, Kroll is now leading an effort to make the team’s model more physiologically relevant by creating a perfusable vascularized kidney organoid-on-chip model.
“This interesting industrial collaboration by Jennifer Lewis’ group wonderfully demonstrates how drug development can be facilitated in a setting where academic research, technology development, and specific therapeutic areas meet and propel each other forward for the betterment of those patient populations who so desperately need new therapies,” said Wyss Founding Director Donald Ingber, M.D., Ph.D., who is also the Judah Folkman Professor of Vascular Biology at Harvard Medical School and Boston Children’s Hospital, and the Hansjörg Wyss Professor of Bioinspired Engineering at the Harvard John A. Paulson School of Engineering and Applied Sciences.
IMAGE CREDIT: Wyss Institute at Harvard University





Leave a Reply