Since it first appeared during the early 1980s, HIV/AIDS has killed an estimated 36.3 million people. While antiretroviral drugs have largely made it a manageable disease in wealthy countries, nearly 1 million people die each year in developing countries.
To date, only a handful of people have been able to completely clear the virus from their system. Each time, it was in a patient who had severe forms of cancer and needed a complete bone marrow transplant. The latest example is a woman from New York. Dr. Carl Dieffenbach from the National Institute of Allergy and Infectious Disease, partial funders of the study, discussed the case with SCINQ.
Can you provide some background regarding the current patient who appears to have cleared HIV from her system and other patients who were “cured” of HIV/AIDS?
A decade or more ago, Timothy Ray Brown, a patient in Germany, was cured of leukemia by bone marrow transplant. His cancer relapsed and he had to be re-induced and got very sick. He ultimately died from his cancer. He was the so-called Berlin patient. He and the London patient had similar courses.
Now, we have the person we are referring to as the New York patient that also had a course of being successfully treated with an HIV antiretroviral therapy, developed a type of leukemia. However, she underwent a different kind of transplant. I think that that is part of the importance of this study. The patient is 17 months out from stopping any antiretroviral therapy and doing quite well.
The reason that type of transplant matters is because she was listed as mixed race – according to the press coverage and the way it was written about by the investigators – and that influences her immunologic makeup. She has genetic markers from Northern Europeans as well as African Americans and that constellation is rare in people who have deposited bone marrow or signed up for bone marrow registries or registries in general.
In this case, the researchers were able to pull together cord blood plus some donor marrow from a relative that allowed the cord blood to be stretched enough that it could successfully lead to a full re-engraftment of what we call a chimera: She’s got now this other individual’s cells and it had the right phenotype of the CCR5 Delta 32 mutation. The rest is all about them stopping therapy and then her progressing to the best possible life living without HIV or taking therapy.
How does the transplant procedure work?
It starts with full body irradiation, a treatment that kills the existing bone marrow. That is a requirement. What you do is you make space or holes in the bone marrow so the
new cells have a place to grow because if you look at a cross section of bone, it is literally packed with tissue. Once that has occurred, they put the transplanted cells in and hope for the best and basically plant the seeds. The cells put themselves into the marrow in the space and take off in an appropriate way and develop into a fully differentiated immune system to a point where now she is not on any other immunosuppressive agents.
That’s one of the issues with some of these other types of transplants. Sometimes you have graft versus host disease or other consequences of receiving a transplant. Apparently, she did not have it.
Different stem cells were used for the Berlin, London, and New York patients. Their first one was from bone marrow. The second was from blood stem cells. This one is umbilical cord stem cells. What are the differences between the three?
The main difference with umbilical cord stem cells is there are so few of them as opposed to other stem cells harvested from marrow. So I think that’s the point of the cord blood and what they call a haploid match. To provide supportive cells that can help the stem cells engraft to a point where they can truly fully function and support the individual’s immune system.
Are they potentially more effective?
I think the issue is that a stem cell is a stem cell. The issue is are they supportive enough? It’s like planting a seed. It has to get comfortable; it has to be able to set down roots; it has to grow and then be able to produce offspring in terms of divide, divide, divide, divide. If there are so few of them, you have fewer (mixed metaphors here) shots on goal to get those cells to take and grow. When you put cord blood into an adult, the issue is just the number of cells.
Do researchers have a decent understanding of how exactly the virus is cleared in this process?
That is one of the interesting things about this is that clearly what we call ablation – the whole body radiation and other antibodies and all that stuff that they do to make space – is a part of it.
In the past, there was a sense that maybe a little graft versus host disease wasn’t such a bad thing and there was some of that that led to additional clearance. In between the different cures, between the Berlin patient, the London patient, there were the Boston patients. They also went through this kind of reduction regimen, ablation, and they were not transplanted with Delta 32. I think they were heterozygous bone marrows, meaning they only had one one allele deleted, and they relapsed after several months to a year or more.
It was clear that the conditioning regimen and the ablation regimen wasn’t sufficient in those individuals, so I’m going to say we don’t know exactly. I think that there are all these different pieces but one of the interesting things that happened with the New York patient is that the patient did not have graft versus host disease so that she managed to make it through without, up to this point, having a relapse again. I don’t think we’re quite ready to say she is cured. I think we still say she’s in remission. I think the sense is we need another year or so before there’s a general sense that maybe she’s completely cured.
Just to clarify, so all of these transplants, this all of the stem cells had that CCR5 mutation on them?
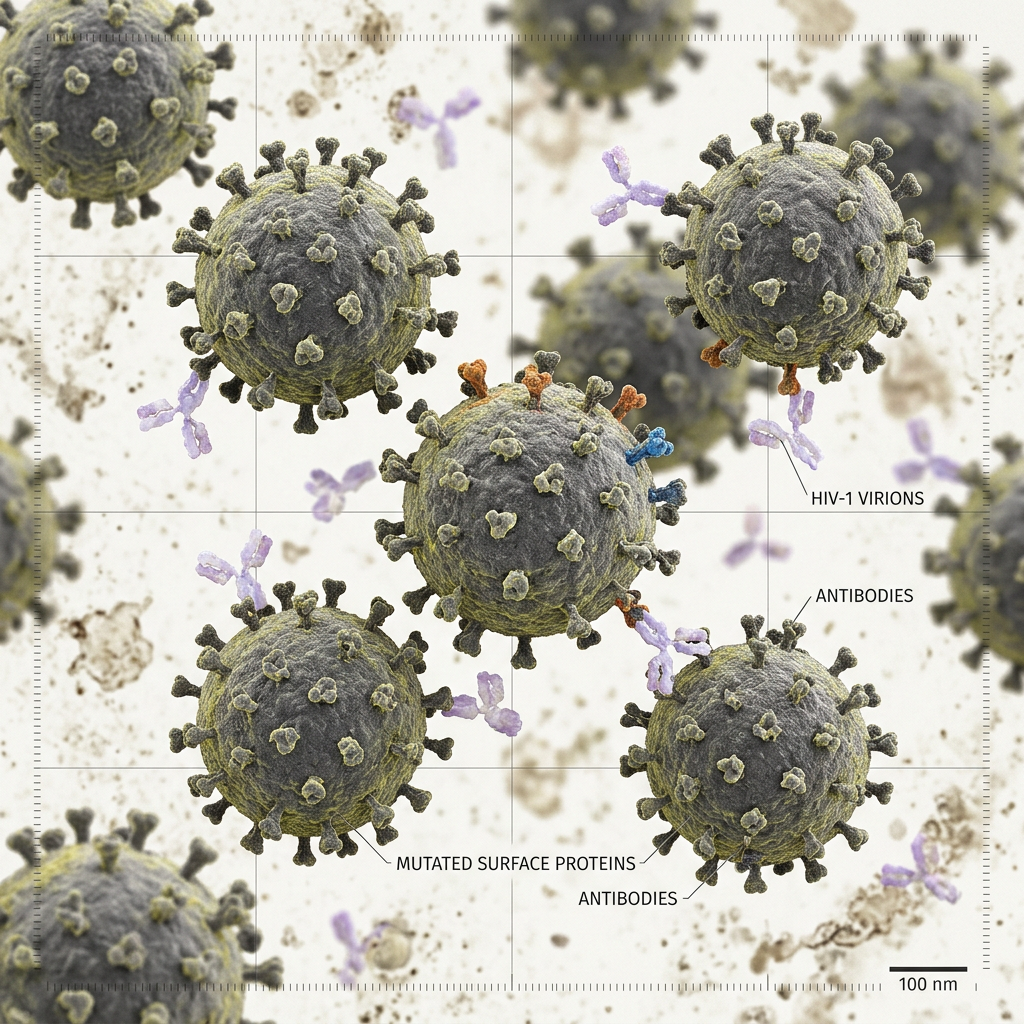
Back in the mid 90s, an HIV co-receptor was discovered. HIV doesn’t enter cells unless it first binds to CD4, and then binds to the co-receptor which is called CCR5. A bunch of different groups discovered that there were people of Northern European descent that lacked the CCR5 and they had a very specific mutation. They were basically resistant to HIV.
That’s where this concept came from of using the bone marrow or cells from a Delta 32 patient as a cure.
Additionally, it provided a new drug target for pharmaceutical companies. Pfizer developed a drug called Maraviroc that blocks CCR5. It went through a variety of very important research findings. There’s a second receptor called CXCR4, but that tends to be used only late in disease as the bone marrow gets wiped out.
The three patients who have gotten HIV into remission all had one had forms of leukemia.
Yes.
So would theoretically would this procedure work on patients without?
It’s not ethical to use this procedure in the absence of disease.
It’s not ethical because the risk is so great. You have to essentially eliminate all their bone marrow and then repopulate it. You only use it in the case where essentially they have an incredibly aggressive cancer that they would die from if they weren’t treated. A bone marrow transplant is the standard of care for AML and for that type of lymphoma that the London patient had. Basically you’re using the standard of care as a two-fer: you’re curing their cancer and also curing their HIV.
So it couldn’t work.
There’s a reason there are only three cures. If you think about it, I mean, there are people that are diagnosed with leukemias all the time. A lot of patients die during this procedure. This leukemia is incredibly aggressive. It’s one of the most aggressive and difficult to treat leukemias. And so this is part of the reason we’ve only seen three.
Okay. So are there other studies similar to these that have gone on that have not worked?
There have been a number of attempts at transplants where the patient has died. There are other strategies that are in development to try to replicate this without a bone marrow transplant. There’s a group in Philadelphia for example, that is using CRISPR technology.
It’s DNA editing. They’re going in with CRISPR and editing patient cells with CRISPR to eliminate the CCR5 gene with the idea of making somebody become Delta 32 through treatment with CRISPR technology. It is a long shot because CRISPR is not the most efficient. It is very efficient, but is it efficient enough?
Do you remember the embryologist in China who got in significant trouble for genetically engineering the human embryos?
Yes.
What he did was use CRISPR to make the embryo cells of those two girls genetically resistant to HIV because he could do it and not for any other reason. And he was, you know, excoriated and kicked out of science.
Basically, it was completely unethical. However, to do it in an adult and not on embryonic tissue is a different matter. According to the FDA and the IRB and all the people at Pitt in Philadelphia, these experiments can go on. So there are ideas for how to use the core information of applying the Delta 32 mutation. Work is being tried to make it more accessible to patients such as use of CRISPR technology.
Does this therapy did this therapy completely? Clear, even latent viruses?
That’s the theory. Yes.
Do any other current therapies come close to what this one does to clearing virus from the body?
Regular antiretroviral therapy — one pill once a day — completely suppresses the growth of the virus in the body, turns it off. There’s no detectable replication. There’s no virus in bodily fluids. You can’t transmit the virus sexually. But it is suppressive. Stop the drugs and the virus comes back. It’s a completely different mechanism and completely different approach.
There are no drugs currently available that go in and actively clear the reservoir of cells in an efficient way that allows you to reduce the size of the reservoir while on therapy. That’s a whole other area of research.
The patients who had gone into remission work were on antiretrovirals is when the therapy started. Does that play any role in the outcome?
I think so. I think it does. The fact that they started all of these individuals started as suppressed and then stayed on therapy during their transplant. That way if there was any residual virus and as parts of the transplant took that were not Delta 32 grew, you could have receded the reservoir.
It was interesting to note that the New York patient was on therapy for three years after the transplant before stopping therapy. They waited a very conservative amount of time before they stopped and decided it was safe for her to stop and watched her for rebounds. And there hasn’t been one.
What is the significance of this? What’s the takeaway, that somewhere down the line, it translates into something tangible?
That’s an incredibly fair and important question. To rephrase your question: So what’s the big deal? And I think that, you know, from a scientific perspective, there’s always new knowledge that is gained from these cases. Part of the new knowledge we gained from this was the use of the cord blood and the Haplo support for the bone marrow graph.
The other interesting thing is there was no graft versus host disease associated in this patient. So you asked that question early on, how was the last little bit of residual reservoir cleared? I think those are all important questions. I think it actually helps to then focus back again on other ways of trying to figure out how to how to do this without a transplant, like with CRISPR or with other genetic technologies to go in and, you know, convert somebody’s bone marrow to Delta 32 or do other techniques to provide genetically protection from reservoir cells that activate. So to me, the study builds and it also bolsters hope that this is an approach that will probably take 10 or 15 years to come to fruition. There’s always something to learn.
Interviewer: Marc Landas.
COPY II (PARAGRAPHS 4 – UNTIL END)
IMAGE CREDIT: (ENTER NAMES)



Leave a Reply