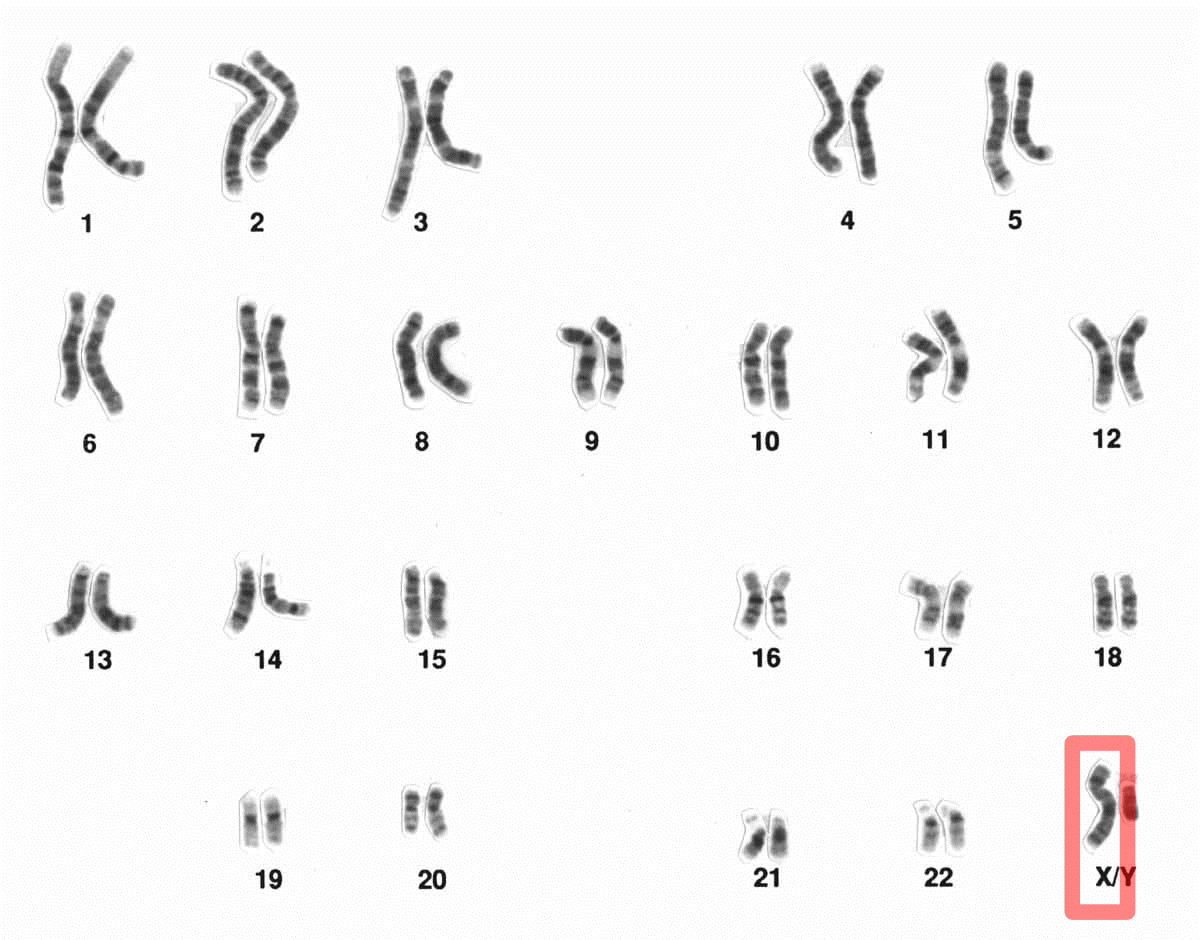
Researchers at Massachusetts General Hospital (MGH) have uncovered new clues that add to the growing understanding of how female mammals, including humans, “silence” one X chromosome. Their new study, published in Molecular Cell, demonstrates how certain proteins alter the “architecture” of the X chromosome, which contributes to its inactivation. Better understanding of X chromosome inactivation could help scientists figure out how to reverse the process, potentially leading to cures for devastating genetic disorders.
Female mammals have two copies of the X chromosome in all of their cells. Each X chromosome contains many genes, but only one of the pair can be active; if both X chromosomes expressed genes, the cell couldn’t survive. To prevent both X chromosomes from being active, female mammals have a mechanism that inactivates one of them during development. X chromosome inactivation is orchestrated by a noncoding form of RNA called Xist, which silences genes by spreading across the chromosome, recruiting other proteins (such as Polycomb repressive complexes) to complete the task.
Jeannie Lee, MD, PhD, an investigator in the Department of Molecular Biology at MGH and the paper’s senior author, has led pioneering research on X chromosome inactivation. She believes that understanding the phenomenon could lead to cures for congenital diseases known as X-linked disorders, which are caused by mutations in genes on the active X chromosome. “Our goal is to reactivate the inactive X chromosome, which carries a good copy of the gene,” says Lee. Doing so could have profound benefits for people with conditions such as Rett syndrome, a disorder brought on by a mutation in a gene called MECP2 that almost always occurs in girls and causes severe problems with language, learning, coordination and other brain functions. In theory, reactivating the X chromosome could cure Rett syndrome and other X-linked disorders.
In this study, Lee and Andrea Kriz, a PhD student and first author of the paper, were interested in understanding the role of clusters of proteins called cohesins in X inactivation. Cohesins are known to play a critical role in gene expression. Imagine a chromosome as a long piece of string with genes and their regulatory sequences being far apart, says Lee. For the gene to be turned “on” and do its job, such as producing a specific protein, it has to come in contact with its distant regulator. Chromosomes allow this to happen by forming a small loop that brings together the gene and regulator. Ring-shaped cohesins help these loops form and stabilize. When the gene’s work is done and it’s time to turn off, a scissor-like protein called WAPL snips it, causing the gene to disconnect from its regulator. An active chromosome has many of these loops, which are continually forming and dissociating (or separating).
These small loops, which are essential for gene expression, are relatively suppressed on an inactivated X chromosome. One reason, as Lee and her colleagues have already shown, is that Xist “evicts” most cohesins from the inactive X chromosome and that this cohesin depletion may be necessary to reorganize the shape and structure of the chromosome for silencing.
In the current study, Lee and Kriz used embryonic stem cells from female mice to find out what happens when cohesin or WAPL levels are manipulated during X chromosome inactivation by using protein-degradation technology. “We found that if cohesin levels build up too high, the X chromosome cannot inactivate properly,” says Lee. Normally, retaining cohesins (which are normally supposed to be evicted) prevented the X chromosome from folding into an inactive shape and gene silencing was affected. “You need a fine balance between eviction and retention of cohesins during X chromosome inactivation,” says Lee.
Next, the authors asked what happens when cohesin is manipulated in an active X chromosome. The short answer: It takes on some peculiar qualities of an inactivated X chromosome. First, when there is insufficient cohesin, the active X develops structures called “superloops” that are usually only seen on the inactive X. Second, when there is too much cohesin, the active X develops “megadomains,” which Lee calls two “big blobs,” and are also ordinarily unique to the inactive X. “The fact that we can confer some features of the inactive X chromosome onto the active X chromosome just by toggling cohesin levels is intriguing,” says Lee. She and her colleagues are trying to understand how and why that happens.
These findings suggests that shape and structure of the X chromosome play a vital role in allowing Xist to spread from one side to the other and achieve inactivation. “The more we learn about what’s important for silencing the X chromosome,” says Lee, “the more likely we’ll be to find ways to reactivate it and to treat conditions like Rett syndrome.”



Leave a Reply