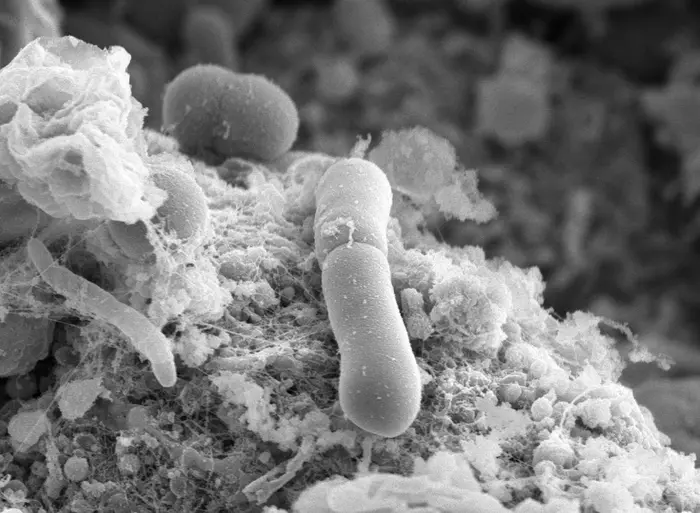
Scientists have discovered how the hospital superbug C.diff rapidly evolves resistance to the frontline drug used for treatment in the UK.
Clostridioides difficile (C. diff), a type of bacteria which often affects people who have taken antibiotics, is responsible for approximately 2,000 deaths annually in the UK.
Researchers from the University of Sheffield and the University of Manchester have found C. diff is able to evolve high levels of vancomycin resistance very quickly – in less than two months the bacteria could tolerate 32 times the normally effective antibiotic concentration.

Currently, the antibiotics used to treat C. diff damage beneficial gut bacteria, leading to a high reinfection rate—up to 30 per cent of patients treated with vancomycin experience a second infection within weeks, with the likelihood of further relapses increasing thereafter.
Despite vancomycin’s critical role within UK healthcare, routine monitoring for resistance in clinical settings is lacking, so resistance may be emerging under the radar in hospitals. If widespread resistance were to arise it would remove this critical treatment option from UK healthcare.
Antimicrobial resistance (AMR) has been identified by the World Health Organisation (WHO) as one of the top global public health and development threats. It is estimated that bacterial AMR was directly responsible for 1.27 million global deaths in 2019 and contributed to 4.95 million deaths.
Sign up for the Daily Dose Newsletter and get every morning’s best science news from around the web delivered straight to your inbox? It’s easy like Sunday morning.
Jessica Buddle, PhD student at the University of Sheffield and lead author of the study, said: “Our findings highlight the need for vigilant monitoring of vancomycin resistance in UK hospitals. Unchecked resistance could contribute to the large number of patients who have a relapsing infection after successful treatment with vancomycin. More research is essential to inform healthcare policy and determine if vancomycin remains the best treatment option.
“Our ongoing work aims to understand the extent and mechanisms of resistance development, simulate these conditions within the complex human gut ecosystem, and collaborate with UK epidemiologists to identify potential resistance signatures in hospitals.
“These efforts are crucial to prevent a future where antibiotics are no longer a viable option for treating bacterial infections and infections that are readily treatable today, become life-threatening once again.”
Although this rapid evolution is concerning, resistant strains exhibited reduced overall fitness, potentially limiting their clinical threat. The resistant strains also commonly had defects in sporulation. Sporulation is essential for C. diff to transmit from one person to the next and to survive on surfaces in hospitals.
Future work will seek to understand this interplay between resistance and the ability of the bacteria to cause severe disease. Researchers will be able to leverage this knowledge to improve surveillance of emerging resistance in hospitals.
IMAGE CREDIT: University of Sheffield
If you enjoy the content we create and would like to support us, please consider becoming a patron on Patreon! By joining our community, you’ll gain access to exclusive perks such as early access to our latest content, behind-the-scenes updates, and the ability to submit questions and suggest topics for us to cover. Your support will enable us to continue creating high-quality content and reach a wider audience.
Join us on Patreon today and let’s work together to create more amazing content! https://www.patreon.com/ScientificInquirer




Leave a Reply