Many of today’s drugs are small, simple molecules. They usually work by regulating the activity of proteins involved in pathologically derailed processes—which is precisely what makes their development extremely complicated. Consequently, a highly adapted molecule has to be developed for each protein, to fit into the corresponding lock—the active center of the protein—like a high-security key. However, proteins actively involved in pathologically derailed processes make up only a fraction of the disease-related proteins. As a result, many proteins are still considered therapeutically “undruggable.”
A majority of the undruggable proteins are compelling targets in cancer research. Perhaps the most prominent among them is the small Ras protein. A single small change in Ras is enough to irreversibly flip the switch for cell growth to “on”—with serious consequences: The cells proliferate rapidly and uncontrollably. Ras mutations occur in nearly a quarter of all tumors. In a groundbreaking study in 2013, a team of researchers led by Herbert Waldmann at MPI in Dortmund developed a new strategy to make Ras, which was previously considered undruggable, druggable: Instead of targeting Ras directly, the researchers used a specially developed molecule to thwart the auxiliary protein PDEd, manipulating the transport and thus the activity of Ras in the cell. However, the researchers did not manage to completely stop the cancer-driving activity of Ras.
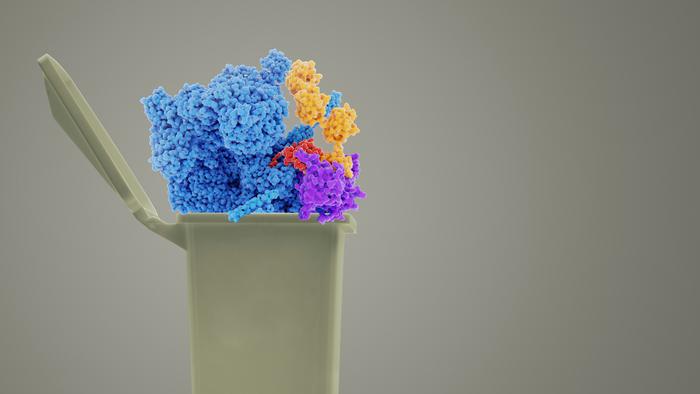
Only two years after Waldmann’s work, American researchers developed a promising new class of drugs to eliminate pathological proteins: they are known as PROTACs (proteolysis-targeting chimeras). These compounds effectively hijack the body’s own protein waste removal system. The large molecule composed of two arms grabs the target protein on one side and the E3 ligase of the protein waste system on the other, which prompts the waste system to dispose of the pathological protein. “That’s an ingenious, truly outstanding scientific achievement,” says Waldmann. “Instead of inhibiting the target protein’s enzymatic activity in a complex process, PROTACs only need to bind to their target with high selectivity. Theoretically speaking, this principle is universally applicable to all proteins, including our Ras transporter PDEd, as we have successfully demonstrated in our current work,” he concludes.

The chemists Waldmann and Winter, along with their teams, created a new PROTAC consisting of the PDEd inhibitor they had developed. They linked the inhibitor to a well-studied molecule that is known to alert another degradation system which can also process larger cell components. “However, our screens have revealed that instead of activating what we call macroautophagy, our PROTAC activates the protein degradation system,” says Georg Winter. He goes on to explain: “What is particularly interesting is that our PROTAC binds a new ligase that was not accessible to the PROTAC strategy so far.”
Currently, there are practically only two E3 ligases that can be used as binding sites for PROTACs. However, there are more than 600 E3 ligases in our bodies. And some of them are only present in very specific tissues. “Tissue-specific ligases could be used to specifically control the site of drug activity,” says Waldmann, looking to the future. “Our rather fortuitous discovery allows for further biological and medicinal-chemical investigation into the ligases we have found. This could help expand the range of pharmaceutically usable PROTACs and, one day, enable the targeted degradation of proteins in specific tissues,” he concludes.
IMAGE CREDIT: MPI of Molecular Physiology
Sign up for the Daily Dose Newsletter and get the morning’s best science news from around the web delivered straight to your inbox? It’s easy like Sunday morning.



Leave a Reply